 Multiple Choice Questions
Multiple Choice QuestionsThe mass of carbon anode consumed (giving only carbondioxide) in the production of 270 kg of aluminium metal from bauxite by the Hall process is (Atomic mass Al= 27)
180 kg
270 kg
540 kg
90 kg
The number of moles of KMnO4 reduced by one mole of KI in alkaline medium is
one-fifth
five
one
two
Which of the following molecules has trigonal planar geometry?
IF3
PCl3
NH3
BF3
D.
BF3
Among all the given options, IF3 has trigonal planar geometry.
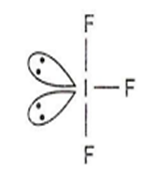
PCl3 has pyramidal geometry.
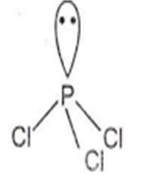
NH3 has pyramidal geometry.
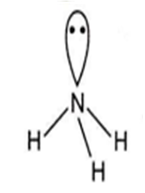
BF3 has trigonal planar geometry
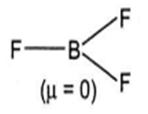
A reaction occurs spontaneously if
TS < H and both H and S are +ve
TS > H and both H and S are +ve
TS = H and both H and S are +ve
TS > H and H is +ve and S is - ve
The correct sequence of increasing covalent character is represented by
LiCl < NaCl < BeCl2
BeCl2 < NaCl < LiCl
NaCl < LiCl < BeCl2
BeCl2 < LiCl < NaCl
Which of the following pairs of a chemical reaction is certain to result in a spontaneous reaction?
Exothermic and decreasing disorder
Endothermic and increasing disorder
Exothermic and increasing disorder
Endothermic and decreasing disorder
A nuclide of an alkaline earth metal undergoes radioactive decay by emission of three -particles in succession. The group of the periodic table to which the resulting daughter element would belong to
Group 14
Group 16
Group 4
Group 6
The absolute enthalpy of neutralisation of the reaction
MgO(s) + 2HCl(aq) MgCl2(aq) + H2O(l)
will be
less than - 57.33 kJ mol-1
- 57.33 kJ mol-1
greater than-- 57.33 kJ mol-1
57.33 kJ mol-1
Equilibrium constants K1 and K2 for the following equilibria
NO (g) + NO2(g) and
2NO2(g) 2NO (g) + O2(g)
are related as
