 Multiple Choice Questions
Multiple Choice QuestionsFrom elementary molecular orbital theory we can deduce the electronic configuration of the singly positive nitrogen molecular ion as :
1s2 , 1s2 , 2s2 , 2s2 , 2p4 , 2p1
1s2 , 1s2 , 2s2 , 2s2 , 2p2 , 2p3
1s2 , 1s2 , 2s2 , 2s2 , 2p3 , 2p2
1s2 , 1s2 , 2s2 , 2s2 , 2p2 , 2p4
The percentage of p-character in the orbitals forming P-P bond in P4 is :
25
33
50
75
D.
75
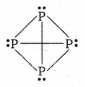 Each P atom in P4 is sp3 hybridised , hence the percentage ofs and p character is 25% and 75% .
Each P atom in P4 is sp3 hybridised , hence the percentage ofs and p character is 25% and 75% .
A solution contains 25% H2O , 25% C2H5OH and 50% CH3COOH by mass .The mole fraction of H2O would be :
0.25
2.5
0.502
5.03
If the solubility of PbCl2 at 25°C is 6.3 x 10-2 mol/L , its solubility product is :
1 x 10-6
1 x 10-3
1.1 x 10-6
1.1 x 10-5
A double bond connecting two atoms .There is a sharing of :
2 electrons
1 electron
4 electrons
All electrons
