 Multiple Choice Questions
Multiple Choice QuestionsThe value of Planck's constant is 6.63 x 10-34 Js. The speed of light is 3 x1017 nms-1 . Which value is closet to the wavelength in nanometer of a quantum of light with frequency of 6 x 1015 s-1 ?
10
25
50
50
What is the maximum number of electrons that can be associated with the following set of quantum number?
n=3, l =1 and m=-1.
10
6
4
4
Based on equation
E=-2.178 x 10-18 J  certain conclusions are written. Which of them is not correct?
certain conclusions are written. Which of them is not correct?
The negative sign in the equation simply means that the energy of an electron bound to the nucleus is lower than if would be if the electrons were at the infinite distance from the nucleus.
Larger the value of n, the larger is the orbit radius
Equation can be used to calculate the change in energy when the electron changes orbit
Equation can be used to calculate the change in energy when the electron changes orbit
How many grams of the concentrated nitric solution should be used to prepare 250 mL o 2.0M HNO3 ? The concentrated acid is 70% HNO3.
45.0 g conc. HNO3
90.0 g conc. HNO3
70.0 g conc. HNO3
70.0 g conc. HNO3
Dipole-induced dipole interactions are present in which of the following pair?
H2O and alcohol
Cl2 and CCl4
HCl and He atoms
HCl and He atoms
A reaction having equal energies of activation for forward and reverse reactions has
6.02 x 1020 molecules of urea are present in 100 mL of its solution. The concentration of solution is
0.02 M
0.01 M
0.001 M
0.001 M
B.
0.01 M
Given, number of molecules of urea =6.02 x 1020
therefore ,Number of moles
= 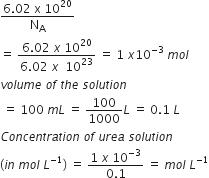
A hydrogen gas electrode is made by dipping platinum wire in a solution of HCl of pH =10 and by passing hydrogen gas through the platinum wire at 1 atm pressure. The oxidation potential of electrode would be
0.0591 V
0.59 V
0.118 V
0.118 V
