 Multiple Choice Questions
Multiple Choice QuestionsThe most suitable reagent for the following conversion, is :
![]()
H2, Pd/C, quinoline
Zn/HCl
Hg2+/H+, H2O
Na/liquid NH3
Which of the following reactions are disproportionation reaction?
(a) 2Cu+ → Cu2+ + Cu0
(b) 3MnO
(c) 2KMnO4
(d) 2MnO
Select the correct option from the following
(a), (b) and (c)
(a), (c) and (d)
(a) and (d) only
(a) and (b) only
For the second period elements the correct increasing order of first ionisation enthalpy is:
Li < B < Be < C < O < N < F < Ne
Li < B < Be < C < N < O < F < Ne
Li < Be < B < C < O < N < F < Ne
Li < Be < B < C < N < O < F < Ne
The number of sigma (σ) and pi (π) pent-2-en-4-yne is
8σ bonds and 5π bonds
11σ bonds and 2π bonds
13σ bonds and no π bonds
10σ bonds and 3π bonds
D.
10σ bonds and 3π bonds
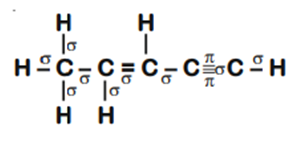
Number of σ bonds = 10 and number of π bonds = 3.
Among the following, the one that is not a green house gas is:
Methane
Ozone
Sulphur dioxide
Nitrous oxide
The method used to remove temporary hardness of water is :
Clark's method
Ion-exchange method
Synthetic resins method
Calgon's method
An alkene "A" on reaction with O3 and Zn–H2O gives propanone and ethanal in equimolar ratio. Addition of HCl to alkene "A" gives "B" as the major product. The structure of product "B" is:
H3C—CH2—CH(CH2Cl)—CH3
![]()
![]()
![]()
Enzymes that utilize ATP in phosphate transfer require an alkaline earth metal (M) as the cofactor. M is :
Mg
Ca
Sr
Be
