 Multiple Choice Questions
Multiple Choice QuestionsTwo small drops of mercury, each of radius R, coalesce to form a single large drop. The ratio of the total surface energies before and after change is
1 : 21/3
21/3 : 1
2 : 1
1 : 2
A black body, at a temperature of 227°C, radiates heat at a rate of 20 cal m-2 s-1. When its temperature is raised to 727°C, the heat radiated by it in cal m-2 s-1 will be closest to
40
160
320
640
C.
320
The temperature of the black body is
T1 = 227°C = 500 K
Stefan-Boltzmann law stated that the total radiant heat energy emitted from a surface is proportional to the fourth power of its absoulute temperature.
∴ Using Stefan's law, the rate of heat radiation per unit area per unit time is
E1 = σT4
⇒ 20 = σ ( 500 )4
⇒ σ =
Now the temperature of the black body is raised to
T2 = 727°C = 1000 K
∴ Rate of heat radiation per unit area
E2 = σ
⇒ = × ( 1000 )4
= 20 × 24
E2 = 320 cal m-2 s-1
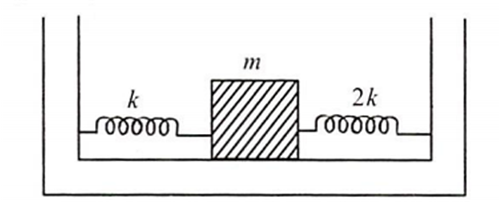
Two springs of force constants k and 2k are connected to a mass as shown in figure . The frequency of oscillation of the mass is
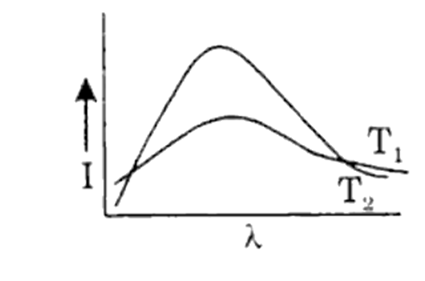
Shown below are the black body radiation curves at temperatures T1 and T2 ( T2 > T1 ). Which of the following plots is correct?



Assertion: Temperatures near the sea coast me moderate.
Reason: Water has a high thermal conductivity.
If both assertion and reason are true and the reason is the correct explanation of the assertion
If both assertion and reason are true and the reason is not the correct explanation of the assertion
If assertion is true but reason is false
If both assertion and reason are false statements
Assertion: When a beetle moves along the sand within a few tens of centimetres of a sand scorpion, the scorpion immediately turns towards the beetle and dashes towards it.
Reason: When a beetle disturbs the sand, it sends pulses along the sand's surface. One set of pulses is longitudinal while the other set is transverse.
if both assertion and reason are true and the reason is the correct explanation of the assertion
if both assertion and reason are true and the reason is not the correct explanation of the assertion
if assertion and reason is false
if both assertion and reason are false statements
Assertion: When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.
Reason: Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours.
if both assertion and reason are true and the reason is the correct explanation of the assertion.
if both assertion and reason are true and the reason is not the correct explanation of the assertion.
if assertion is true but reason is false
if both assertion and reason are false statements
Assertion: The size of a hydrogen balloon increases as it rises in air.
Reason: The material of the balloon can be easily stretched.
if both assertion and reason are true and the reason is the correct explanation of the assertion
if both assertion and reason are true and the reason is not the correct explanation of the assertion
if assertion is true but reason is false
if both assertion and reason are false statements
Assertion: It is hotter over the top of a fire than at the same distance on the sides
Reason: Air surrounding the fire conducts more heat upwards.
if both assertion and reason are true and the reason is the correct explanation of the assertion
if both assertion and reason are true and the reason is not the correct explanation of the assertion
if assertion is true but reason is false
if both assertion and reason are false statements
Assertion: The amplitude of an oscillating pendulum decreases gradually with time.
Reason: The frequency of the pendulum decreases with time.
if both assertion and reason are true and reason is the correct explanation of assertion
if both assertion and reason are true and the reason is not the correct explanation of the assertion
if assertion is true but reason is false
if both assertion and reason are false statements
