
B.
the fuel is not burning completelyA homologous series is a group of compounds which contains the same functional group but different chain lengths. Thus, these have the same chemical properties but different physical properties.
Example:
CH3OH
C2H5OH
C3H7OH
C4H9OH
Above are members of the same homologous series having different chain length and same functional group.
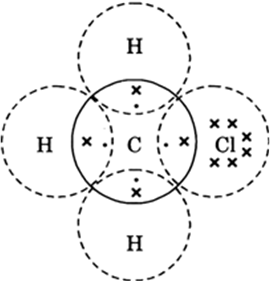
Explain the nature of the covalent bond using the bond formation in CH3Cl.
|
Teat |
Ethanol |
Ethanoic acid |
|
1. Litmus test 2. Sodium bicarbonate 3. Sodium metal test |
No change in colour of litmus solution. No brisk effervescence. H2 is given out with effervescence. |
Blue litmus solution turns red. Brisk effervescence due to evolution of CO2. H2 is produced but no effervescence. |
