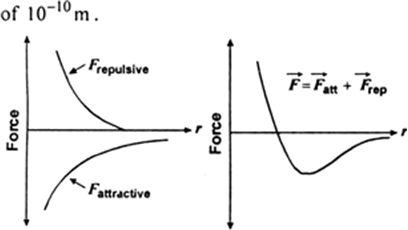
Amorphous substances do not have sharp melting point but get softening range before melting, while crystalline substances have sharp melting point. Explain.
Amorphous substances possess short-range arrangement of atoms or molecules. These short and ordered arrangement disappears gradually on heating. Before melting, this gradual change makes the amorphous substance soft.
While in crystalline substances, there is a long-range order arrangement of atoms or molecules. This arrangement disappears at a definite temperature and hence have sharp melting points.
291 Views