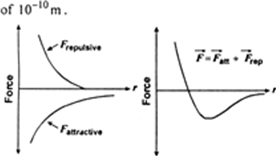
A rectangular beam is bent into arc of a circle. What type of strain is produced in it?
When rectangular beam is bent into arc of a circle, the layers on convex side of neutral plane undergo extensional stress and the layers that are on concave side undergo compressional stress.
252 Views