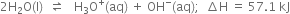What are the limitations of Lewis concept of acids and bases ?
(i) The concept fails to recognise the relative strength of acids and bases.
(ii) Formation of a coordination compound is a slow process but acid-base reactions are fast. This concept could not explain this behaviour.
(iii) Protonic acids (H2SO4, HCl, HNO3) do not form a coordinate bond with bases and therefore are not included in the Lewis acids.
(iv) The catalytic activity of many acids is due to H+ ion. Since a Lewis acid need not contain hydrogen hence many Lewis acids will not have catalytic activity.
What is the effect of temperature on pH?
We know that ionic product of water (Kw.) increases with the rise of temperature. Therefore, the concentration of H3O+ ions increases with temperature.
Therefore, pH value is inversely proportional to  ion concentration
ion concentration
Hence pH value decreases with the rise in temperature.
Out of Lowry Bronsted concept and Lewis concept, which is regarded as better and why?
Lewis concept of acids and basis is regarded as better than Bronsted concept due to the following reasons:
(i) Many species which are acids and bases according to Bronsted and Arrhenius concept are also covered by Lewis concept.
(ii) The acidic and basic character of certain species can be explained only with the help of Lewis concept. For example, acidic nature of CO2 and basic nature of CaO can not be explained by any other concept except the Lewis concept.
(iii) According to Lewis concept, an acid need not contain the element hydrogen but it must be electron deficient in nature. For example AlCl3 is an acid though it has no H+ion present.
The ionic product of water is 0·11 × 10–14 at 273 K; 1·0 × 10–14 at 298K and 51 × 10–14 at 373K. Deduce from this data whether the ionisation of water into hydrogen and hydroxide ion is exothermic or endothermic.