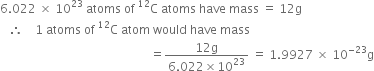In a reaction![]()
Identify the limiting reagent, if any, in the following reaction mixtures.
(i) 300 atoms of A + 200 molecules of B
(ii) 2 mol A + 3 mol B
(iii) 100 atoms of A + 100 molecules of B
(iv) 5 mol A + 2-5 mol B
(v) 2·5 mol A + 5 mol B
According to the given reaction:
(i) 1 atom of A will react with molecules of B2 = 1
300 atoms of A will react with molecules of B2 = 300
But molecules of B2 actually available = 200
∴ B2 is the limiting reactant.
(ii) 1 mol of A reacts with 1 mol of B
∴ 2 mol of A will react with 2 mol of B.
Hence A is the limiting reactant.
(iii) 100 atoms of A will react with 100 molecules of B. Hence there are no limiting reactants.
(iv) 2·5 mol of B will react with 2·5 mol of A. Hence B is the limiting reagent.
(v) 2·5 mol of A will react with 2·5 mol of B.
Hence A is the limiting reagent.
How are 0.50 mol Na2CO3 and 0·50 M Na2CO3 different?
Molar mass of Na2CO3 = 2 x 23 + 12 + 3 x 16 = 106 g
0.50 mol Na2CO3 means 0.50 x 106g = 53g
0.50M Na2CO3 means 0.50 mol i.e. 0.50 x 106g, i.e. 53 g of Na2CO3 are present in 1 litre of the solution.
Calculate the molarity of a solution of ethanol in water in which the mole fraction of ethanol is 0.040.(assume the density of water to be one).
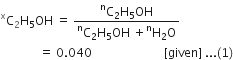
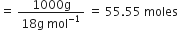
 in eq. (1) we have
in eq. (1) we have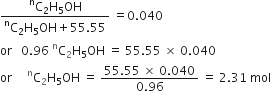
What will be the mass of one ![]() atom in g?
atom in g?
1 mol of ![]() atoms =
atoms = 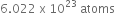 = 12 g
= 12 g