Define the term significant figures.
Significant figures are meaningful digits which are known with certainty.
What weight of Ca will contain the same number of atoms as are present in 3.2g of sulphur?
Atomic mass of sulphur = 32 amu
Gram atomic mass of sulphur = 32 g
1 mole of sulphur contains = 6.023 x 1023 atoms
Now 32 g of sulphur contains
= 6.023 x 1023 atoms
3.2 g of sulphur would contain
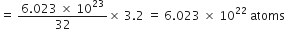
Gram atomic mass of calcium = 40 g
1 mole of calcium = 40 g
= 6.023 x 1023 atoms
Now 6.023 x 1023 atoms of calcium have mass = 40 g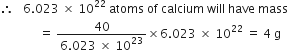
Express the Avogadro's number (6·023×1023) upto three significant figures.
