A gas occupies a volume of 300 mL at 740 mm Hg at 20°C. What additional pressure is required to reduce the gas volume to 250 ml at 20°C?
From the given data.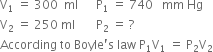
Substituting the values in the above equation,

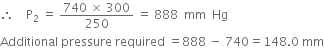
What pressure must be applied to a given sample of a gas in order to compress it to three-fourth of its original volume ?
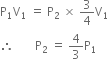 times the original pressure.
times the original pressure.
Is it possible to cool a gas below absolute zero?
Why is kelvin scale of temperature regarded better than centigrade scale?
