Calcualte the energy of one mole of photons of radiation whose frequency is 5 x 1014Hz.
Energy (E) of one photon = hv
Here, h = 6.626 x 10-34 Js
v = 5 x 1014 s-1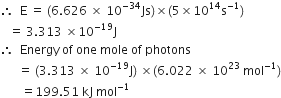
Here, 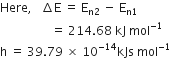
According to Planck's quantum theory,
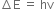
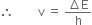 ....(1)
....(1)
Substituting the value in (1),
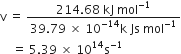
Energy (E) of one photon = hv =  ...(1)
...(1)
Here 
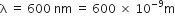
Substituting the values in eq. (1), we have
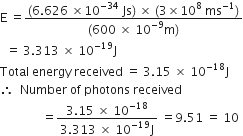
Application of Quantum theory:
(i) In 1905, Einstein applied this theory to explain the photoelectric effect.
(ii) In 1913, N. Bohr used this theory to explain the structure of atoms and explain hydrogen spectra.
(iii) In 1922, this theory was used to explain Compton effect.
