Write the complete symbols for the nuclei having:
(i) atomic number 26 and mass number 55,
(ii) atomic number 4 and mass number 9
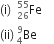
Atomic number is defined in terms of protons and not in terms of electrons. Why?
Calculate the number of neutrons in dipositive zinc ion (Zn2+, the mass number being 70).
No. of neutrons
= Mass number - Atomic number
Atomic mass of zinc is 70 g
Atomic number of Zinc is 30. Thus, number of neutrons will be,
No. of neutrons = 70 - 30 = 40
