Why do the following reactions proceed differently?
Pb3O4 +8HCl --->3PbCl2 +Cl2 +4H2O
Pb3O4 +4HNO3 --->2Pb(NO3)2 +PbO2 +2H2O
Determine the oxidation number of C in the following: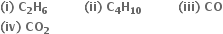
C2H6: Let the oxidation number of C= x
Oxidation number of H = 1
The algebraic sum of the oxidation number of various atoms = 0
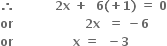
 4x + 10(+1) = 0
4x + 10(+1) = 0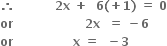
 x - 2 = 0
x - 2 = 0 x + 2(-2) = 0
x + 2(-2) = 0What is the oxidation number of sulphur in:
(i)H2SO4 (ii) Na2S2O3?
(i) H2SO4:
Let the oxidation number of S = x
Writing the oxidation number of each atom at the top of its symbol
+1 x -2
H2 S O4
The algebraic sum of the oxidation number of various atoms = 0
2(+1) + x + 4(-2) = 0
x - 6 = 0
x = +6
(ii) Na2S2O2:
Let the oxidation number of S = x
Writing the oxidation number of each atom at the top of its symbol.
+1 x -2
Na2 S2 O3
The algebraic sum of the oxidation number of various atoms = 0
2(+1) + 2(x) + 3(-2) = 0
or 2 + 2x - 6 = 0 x = +2
x = +2
Determine the oxidation number of:
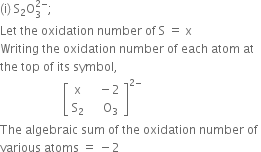
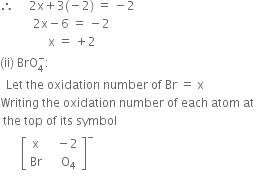
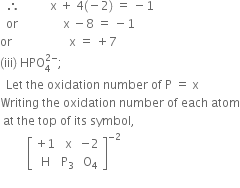
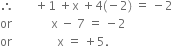
ClO- (hydrochlorite ion):![]()
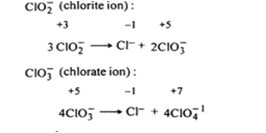
ClO4-1 (perchlorate ion) cannot show any disproportionation reaction. The oxidation state of Cl is ClO4- ion is +7. It is the maximum oxidation state which it can have. It is the maximum oxidation state which it can have. It can decrease the same by undergoing reduction and not increase it anymore hence ClO4- ion does not undergo disproportionation reactions.
