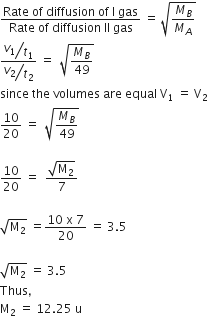In Duma's method of estimation of nitrogen 0.35 g of an organic compound gave 55 mL of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound would be
(Aqueous tension at 300 k = 15 mm)
16.45
17.45
14.45
14.45
A.
16.45
Gas equation,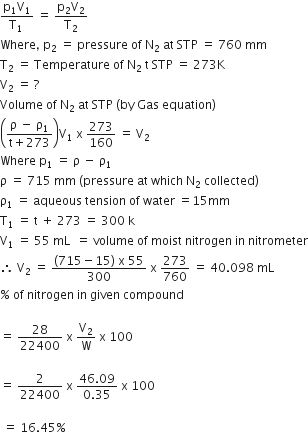
A gas such as carbon monoxide would be most likely to obey the ideal gas law at
high temperatures and low pressures
low temperatures and high pressures
high temperatures and high pressures
high temperatures and high pressures
A.
high temperatures and low pressures
Real gases show ideal gas behaviour at high temperatures and low pressures.
PV = nRT
By what factor does the average velocity of a gaseous molecule increase when the temperature (in kelvin) is doubled?
2.8
4.0
1.4
1.4
C.
1.4
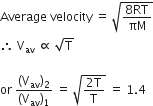
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 seconds respectively. The molecular mass of A is 49u. Molecular mass of B will be
12.25 u
6.50 u
25.00 u
25.00 u
A.
12.25 u