we have given that
Ecell0 =0.36V
thus equilibrium constant is
n=2
k =1.596 x 1012
Definition: The standard hydrogen electrode is the standard measurement of electrode potential for the thermodynamic scale of redox potentials.
The standard is determined by the potential of a platinum electrode in the redox half reaction
2 H+(aq) + 2 e- → H2(g) at 25 °C.
The standard hydrogen electrode is often abbreviated SHE.
Also Known As: normal hydrogen electrode or NHE
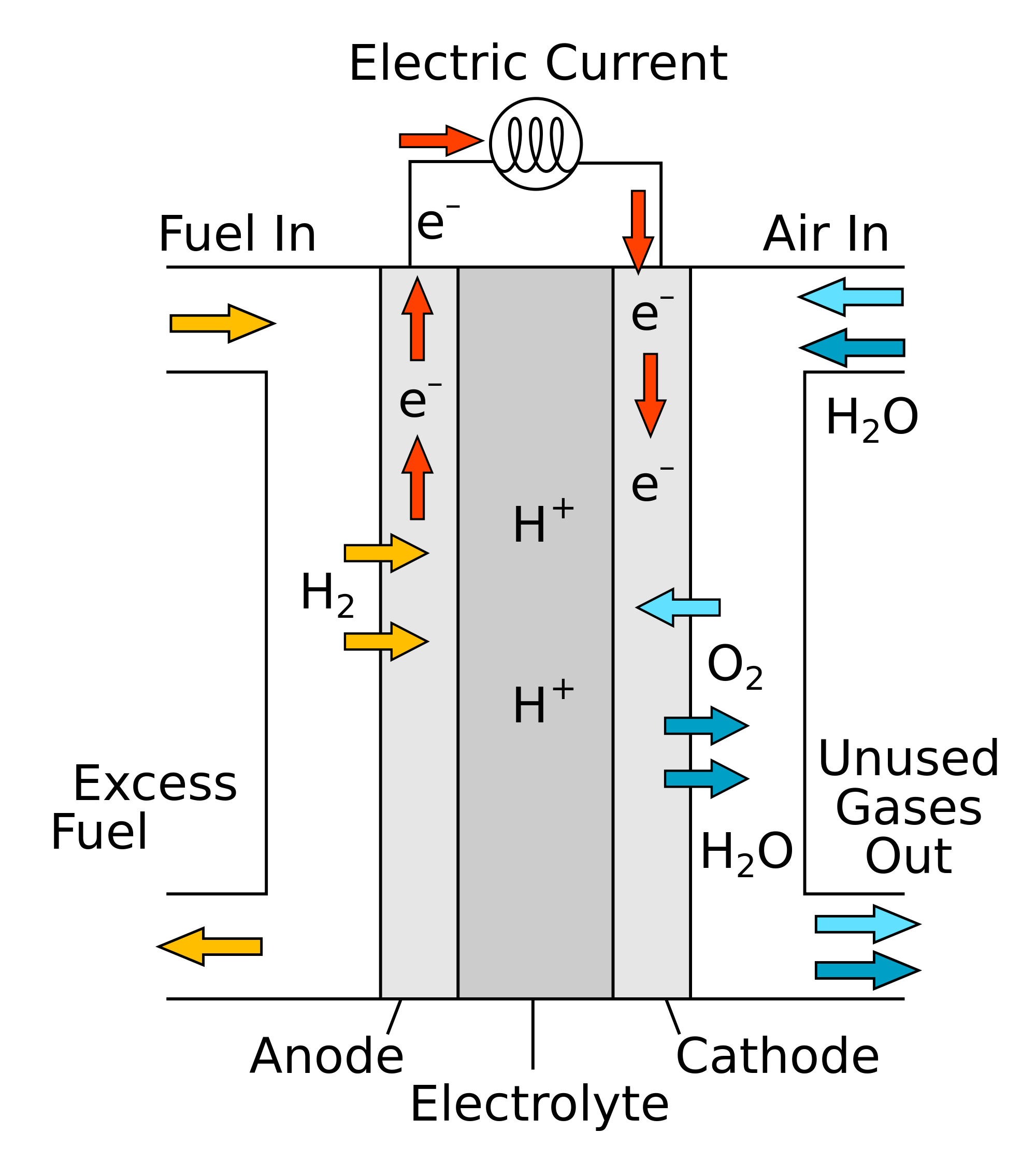
On the basis of the standard electrode potential values stated for acid solution. Predict whether Ti4+ species may be used to oxidise FeII to FeIII.
Reaction: E°/V TiIV + e– → Ti3+ + 0.01
Fe3+ + e– → Fe2+ + 0.77
Ti4+ would not oxidize Fe+2 . since the standard electrode potential of iron is high as compare to the titanuim standard electrode potential.
Predict the products of electrolysis obtained at the electrodes in each case when the electrodes used are of platinum.
(i) An aqueous solution of AgNO3.
(ii) An aqueous solution of H2SO4.
