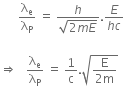A caesium photocell, with a steady potential difference of 60 V across it, is illuminated by a bright point source of light 50 cm away. When the same light is placed 1 cm away, the photoelectrons emitted from the cell
are half as numerous
are one quarter as numerous
each carry one quarter of their previous energy
each carry one quarter of their
B.
are one quarter as numerous
Number of photo electrons emitted will be 1/4th as previous.
When a metallic surface is illuminated with radiation of wavelength  , the stopping potential is V. If the same surface is illuminated with radiation of wavelength 2
, the stopping potential is V. If the same surface is illuminated with radiation of wavelength 2  , the stopping potential is V/4. The threshold wavelength for the metallic surface is,
, the stopping potential is V/4. The threshold wavelength for the metallic surface is,


3
3
C.
3
When a metallic surface is illuminated with radiation of wavelength  , the stopping potential is V.
, the stopping potential is V.
Photoelectric equation can be written as,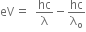 ... (i)
... (i)
Now, when the same surface is illuminated with radiation of wavelength 2 , the stopping potential is V/4. So, photoelectric equation can be written as,
, the stopping potential is V/4. So, photoelectric equation can be written as,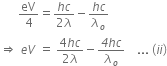
From equations (i) and (ii), we get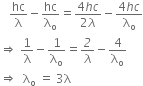
A radiation of energy 'E' falls normally on a perfectly reflecting surface. The momentum transferred to the surface is
(c = velocity of light)

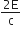
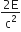
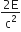
B.
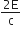
The radiation energy is given by 
A black body is at a temperature of 5760 K. The energy of radiation emitted by the body at wavelength 250 nm is U1, at wavelength 500 nm is U2 and that at 1000 nm is U3. Wien's constant, b = 2.88 x 106 nmK. Which of the following is correct?
U3 = 0
U1 > U2
U2 > U1
U2 > U1
C.
U2 > U1
Given, temperature, T1 = 5760 K
Given that energy of radiation emitted by the body at wavelength 250 nm in U1, at wavelength 500 nm is U2 and that at 1000 nm is U3.
Now, according to Wein's law, we get
where, b = Wien's constant = 2.88 x 106 nmK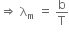

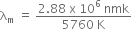

 is the wavelength corresponding to maximum energy, so U2 > U1.
is the wavelength corresponding to maximum energy, so U2 > U1.
An electron of mass m and a photon have the same energy E. The ratio of de-Broglie wavelength associated with them is,
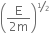

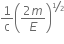
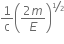
D.
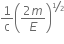
Given that electron has a mass m.
De-Broglie wavelength for an electron will be given as,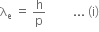
where,
h is the Planck's constant, and
p is the linear momentum of electron
Kinetic energy of electron is given by, E = 

From equation (i) and (ii), we have
Energy of a photon can be given as,
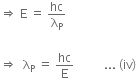
Hence,  is the de-Broglie wavelength of photon.
is the de-Broglie wavelength of photon.
Now, dividing equation (iii) by (iv), we get