A salt bridge is a U-shaped device containing concentrated solution of an inert electrolyte like KCl, KNO3, etc.or a solidified solution of those electrolytes in agar-agar solution and gelatin. It connects the oxidation and reduction half-cells of a galvanic cell. The inert electrolytes present do not take part in redox reaction of the cell and dont react with the electrolyte that has been used.
As electrons leave one half of a galvanic cell and flow to the other, a difference in charge is built up. If no salt bridge were used, this increasing charge difference would eventually prevent further flow of electrons. The salt bridge solves these problems. It has two main functions:
1. To allow the flow of ions from one solution to another without mixing of the two solutions and completing the electrical circuit.
2. To maintain the electical neutrality of the solutions in the two half cells.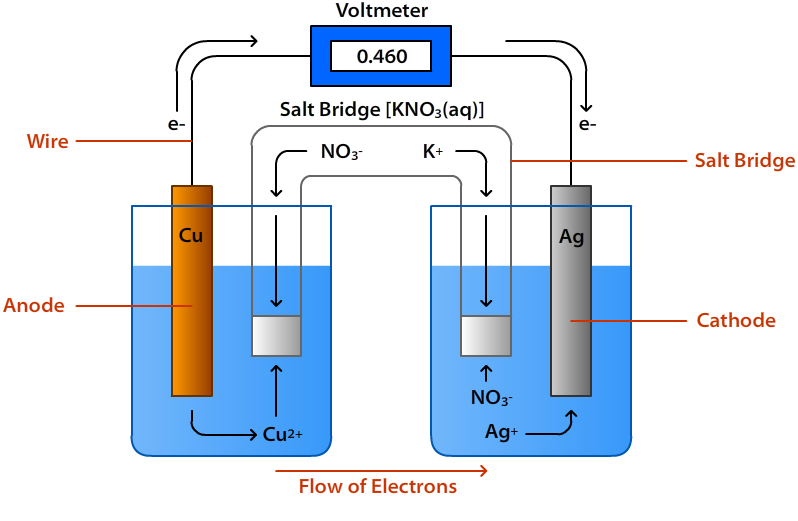
The resistance of any conductor varies directly as its length (l) and inversely as its cross-sectional area (A), i.e.,
Mathematically
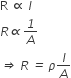
Where ρ is called the specific resistance.
l/A is known as cell constant.
If l = 1 cm and A = 1 cm2, then
R = ρ
The specific resistance is, thus defined as the resistance of one-centimetre cube of a conductor.
The reciprocal of specific resistance is termed the specific conductance or it is the conductance of one-centimetre cube of a conductor.
It is denoted by the symbol κ.
Thus, 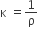
Where (κ) kappa → the specific conductance
Specific conductance is also called conductivity.
Further,
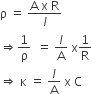
or Specific conductance = Conductance × cell constant
In the case of electrolytic solutions, the specific conductance is defined as the conductance of a solution of definite dilution enclosed in a cell having two electrodes of unit area separated by one centimetre apart.
Unit of specific conductance: Ω-1 cm-1
It is not possible to store copper sulphate solution in iron vessel.since ,iron is more reactive than copper,it displaces copper from any if its solution.the reaction takes place as ,
Fe(s)+CuSO4(aq)..............>FeSO4(aq)+Cu(s)
Free energy change G is measure of the spontaneity of a chemical reaction or process.
Where
-n =number of moles of electron
F= the quantity of electrical charge that is contained in 1mole of electrons.This is the Faraday constant i.e. 1F= 96500 Coloum/mole of electron
F and n are positive value. therefore as positive value of E (which indicte spontaneity )and thus negtive value for G (which also indicate spontaneity)
