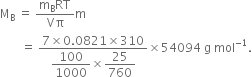Heptane and Octane form an ideal solution at 373 K. The vapour pressures of the pure liquids at this temperature are 105.2 K Pa and 46.8 K Pa respectively. If the solution contains 25 g of heptane and 28.5 g of octane, calculate
(i) Vapour pressure exerted by heptane.
(ii) Vapour pressure exerted by solution.
(iii) Mole fraction of octane in the vapour phase.
Components A (Heptane, )
Components of B
No. of moles of heptane,
No. of moles of octane,
Total moles in solution,
(i) Vapour pressure exerted by heptane
where is mole fraction of component A
(ii) Vapour pressure of octane
Total vapour pressure of solution
(iii) Mole fraction of octane
Osmotic pressure: Osmotic pressure is the minimum pressure that should be applied to the more concentrated solution to prevent osmosis.
Determination of osmotic pressure: Barkley and Hartley’s method: The apparatus consists of a porous pot containing copper ferrocyanide deposited in its wall (acts as semipermeable membrane) and fitted into a bronze cylinder to which is fitted a piston and a pressure gauge (to read the applied pressure).
The pot is fitted with a capillary indicator on left and water reservoir on right. Pot is filled with water while the cylinder is filled with a solution whose osmotic pressure is to be measured. Water tends to pass into the solution through the semipermeable membrane with the result that the water level in the indicator falls down. External pressure is now applied with piston so as to maintain a constant level in the indicator. This external pressure is osmotic pressure.
If the membrane used was a slightly, leaky, then the measured valued of osmotic pressure will not be definite.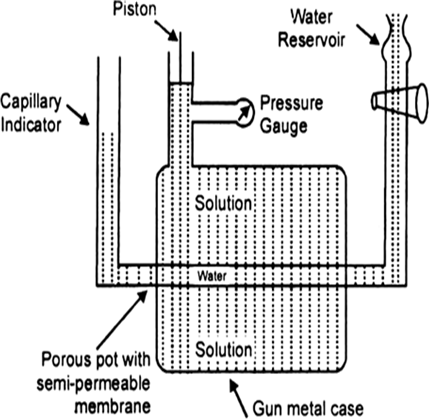
Fig. Barkley and Hartley’s apparatus.
(i) Volume of solution = 1 L = 1000 mL
Density of solution = 1.84 g mL–1
Mass of solution = V x d
= (1000 mL) x 1.84 mL–1
= 18540 g
Mass of H2SO4 = 93g/100mL
93 x 10 = 930 g
Mass of solvent (water)
= 1840 – 930 = 910 g.
(ii) Molarity (m)
(a) Measurement of Osmotic Pressure. Different methods are employed for the measurement of osmotic pressure in the laboratory but Berkley and Hartley's method gives the best results. The apparatus consists of a porous pot containing copper ferrocyanide deposited in its wall (acts as semi-permeable membrane) and fitted into a bronze cylinder to which is fitted a piston and a pressure gauge (to read the applied pressure).
The pot is fitted with a capillary indicator on left and water reservoir on right. Pot is filled with water while the cylinder is filled with a solution whose osmotic pressure is to be measured. Water tends to pass into the solution through the semipermeable membrane with the result that the water level in the indicator falls down. External pressure is now applied with piston so as to maintain a constant level in the indicator. This external pressure is osmotic pressure.
If the membrane used was a slightly, leaky, then the measured valued of osmotic pressure will not be definite.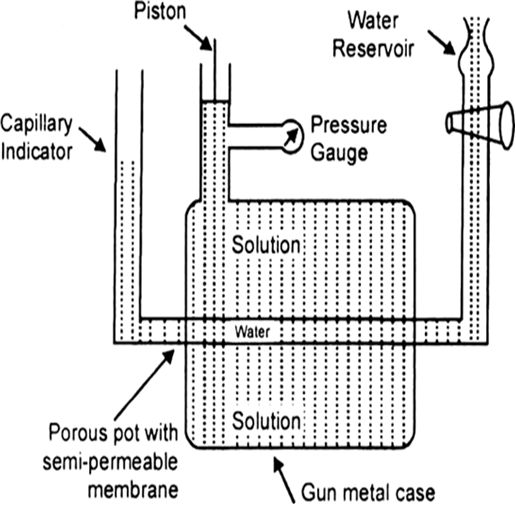
Fig. Berkley and Hartley's apparatus.
(b)