In case of positive deviation from Raoult's law, A-B interactions are weaker than those between A-A or B-B, i.e., in this case the intermolecular attractive forces between the solute-solvent molecules are weaker than those between the solute-solute and solvent-solvent molecules. This means that in such solutions, molecules of A (or B) will find it easier to escape than in pure state. This will increase the vapour pressure and result in positive deviation. Mixtures of ethanol and acetone is good example of this.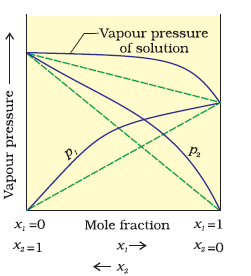
Answer:
osmotic pressure is proportional to the molarity, C of the solution at a given temperature T. Thus:
Π = C R T
Here Π is the osmotic pressure and R is the
gas constant.
Π = (n2 /V) R T
Here V is volume of a solution in litres containing n2 moles of solute.
If w2 grams of solute, of molar mass, M2 is present in the solution, then
n2 = w2 / M2 and we can write,
Thus, knowing the quantities w2, T, Π and V we can calculate the molar mass of the solute.
Answer:
the vapour pressure of a liquid increases with increase of temperature. It boils at the temperature at which its vapour pressure is equal to the atmospheric pressure. For example, water boils at 373.15 K (100° C) because at this temperature the vapour pressure of water is 1.013 bar (1 atmosphere)
Let be the boiling point of pure solvent and
Tb be the boiling point of solution. The increase in
the boiling point is known as
elevation of boiling point.
for dilute solutions the elevation of boiling point (ΔTb) is directly proportional to the molal concentration of the solute in a solution. Thus
ΔTb ∝ m
or ΔTb = Kb m
Here m (molality) is the number of moles of solute dissolved in 1 kg of solvent and the constant of proportionality, Kb is called Boiling Point Elevation Constant or Molal Elevation Constant (EbullioscopicConstant). The unit of Kb is K kg mol-1. If w2 gram of solute of molar mass M2 is dissolved in w1 gram of solvent, then molality, m of the solution is given by the expression:
Thus, in order to determine M2, molar mass of the solute, known mass of solute in a known mass of the solvent is taken and ΔTb is
determined experimentally for a known solvent whose Kb value is known.
Answer:
Elevation of boiling point with addition of non-volatile solute vapour pressure decrese and hence boiling point increase.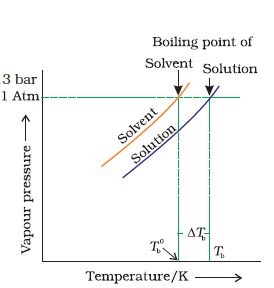
Let be the boiling point of pure solvent and
Tb be the boiling point of solution. The increase in
the boiling point is known as
elevation of boiling point.
for dilute
solutions the elevation of boiling point (ΔTb) is
directly proportional to the molal concentration of
the solute in a solution. Thus
ΔTb ∝ m
or
ΔTb = Kb m
Here m (molality) is the number of moles of solute dissolved in 1 kg of solvent and the constant of proportionality, Kb is called Boiling Point Elevation Constant or Molal Elevation Constant. The unit of Kb is K kg mol-1.If w2 gram of solute of molar mass M2 is dissolved in w1 gram of solvent, then molality, m of the solution is given by the expression:
Thus, in order to determine M2, molar mass of the solute, known mass of solute in a known mass of the solvent is taken and ΔTb is
determined experimentally for a known solvent whose Kb value is known.
