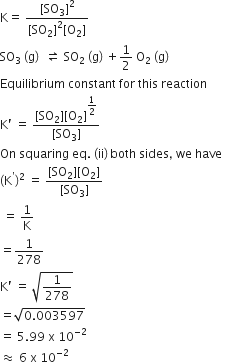Buffer solutions have constant acidity and alkalinity because
these give unionised acid or base on reaction with added acid or alkali
acids and alkalies in these solutions are shielded from attack by other ions.
they have a large excess of H+ or OH- ions
they have a large excess of H+ or OH- ions
A.
these give unionised acid or base on reaction with added acid or alkali
If a small amount of an acid or alkali is added to a buffer solution, it converts them into unionised acid or base. Thus, remains unaffected or in other words its acidity/alkalinity remains constant. e.g.,
H3O+ + A- ⇌ H2O +HA
-OH +HA → H2O +A-
If acid is added, it reacts with A- to form undissociated HA. Similarly, if base/alkali is added, O H- combines with HA to give H2O and A- and thus, maintains the acidity/alkalinity of buffer solution.
Given the reaction between two gases represented by A2 and B2 to give the compound AB (g).
A2(g) +B2 (g) ⇌ 2AB (g)
At equilibrium the concentration
of A2 = 3.0 x 10-3 M
of B2 = 4.2 x 10-3 M
of AB = 2.8 x 10-3 M
If the reaction takes place in a sealed vessel at 527oC, then the value of Kc will be
2.0
1.9
0.62
0.62
C.
0.62
A2 (g) +B2 (g) ⇌ 2AB (g)
The equilibrium constant is given by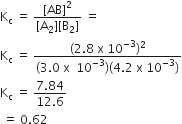
Equimolar solutions of the following substances were prepared separately, which one of these will record the highest pH value?
BaCl2
AlCl3
LiCl
LiCl
A.
BaCl2
BaCl2 is a salt of strong acid HCl and strong base Ba(OH)2. So, its aqueous solution is neutral with pH 7. All other salts give acidic solution due to cationic hydrolysis, so their pH is less than 7. Thus, pH value is highest for the solution of BaCl2.
pH of a saturated solution of Ba(OH)2 is 12. The value of solubility product Ksp of Ba(OH)2 is
3.3 x 10-7
5.0 x 10-7
4.0 x 10-6
4.0 x 10-6
B.
5.0 x 10-7
Given, pH of Ba(OH)2 = 12
pOH = 14-pH
= 14-12 = 2
We know that,
pOH = -log [OH-]
2 =-log [OH-]
[OH-] = antilog (-2)
[OH-] = 1 x 10-2
Ba(OH)2dissolves in water as 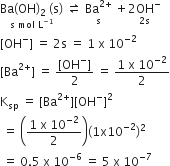
Give that the equilibrium constant for the reaction,
2SO2 (g) + O2 (g) ⇌ 2SO3 (g)
has a value of 278 at a particular temperature. What is the value of the equilibrium constant for the following reaction t the same temperature?
SO3 (g) ⇌ SO2 (g) +1/2 O2 (g)
1.8 x 10-3
3.6 x 10-3
6.0 x 10-2
6.0 x 10-2
C.
6.0 x 10-2
2SO2 (g) +O2 (g) ⇌ 2SO3 (g)
Equilibrium constant for this reaction,