
Try segregating the things around you as pure substances or mixtures.
Pure substances—copper wire, water, sugar, salt, etc.
Mixtures—fruits, furniture, paper, clothes, food, butter, shoes, vegetables, toothpaste, etc.
Name the technique to separate:
(i) butter from curd,
(ii) salt from sea-water
(iii) camphor from salt.
(i) Butter from curd = centrifugation
(ii) Salt from sea-water = crystallization
(iii) Camphor from salt = Sublimation
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than  ), which are miscible with each other?
), which are miscible with each other?
We can separate a mixture containing kerosene and petrol by simple distillation because kerosene and petrol do not decompose on heating and their boiling points are sufficiently apart. The apparatus used for the simple distillation is given below.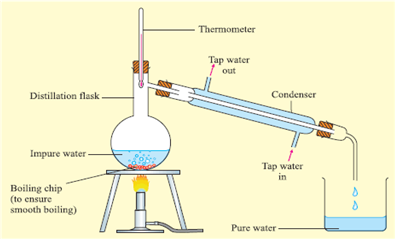
What type of mixtures are separated by the technique of cyrstallisation?
Crystallization is used to purify salt that we get from sea water and to obtain pure substance from their impure samples.
Classify the following as chemical or physical changes:
• cutting of trees
• melting of butter in a pan
• rusting of almirah
• boiling of water to form steam
• passing of electric current through water and the water breaking down into hydrogen and oxygen gases
• dissolving common salt in water
• making a fruit salad with raw fruits
• burning of paper and wood.
• Cutting of trees—physical change
• melting of butter in a pan—physical change
• rusting of almirah—chemical change
• boiling of water to form steam—physical change
• passing of electric current through water and the water breaking down into hydrogen and oxygen gases—chemical change
• dissolving common salt in water—physical change
• making a fruit salad with raw fruits—physical change
• burning of paper and wood—chemical change
