 Long Answer Type
Long Answer Typea) Give a chemical test to distinguish between saturated and unsaturated hydrocarbon.
b) Name the products formed when ethane burns in the air. Write a balanced chemical equation for the reaction showing the types of energies liberated.
c) Why is the reaction between methane and chlorine in the presence of sunlight considered a substitution reaction?
A chemical test to distinguish between saturated and unsaturated hydrocarbon.
Bromine water test - Unsaturated hydrocarbon gives addition reaction with Br2. So the colour of Br2-water gets decolourized.
R-C=C=R + Br2 --> R-BrC-CBr-R
whereas,
Saturated hydrocarbons do not react with Br2-water so the colour of Br2 -water does not get decolourised.
b) when ethane (CH3-CH3) is Burned in air (oxygen), it forms carbon dioxide and water.
CH3-CH3 +7/2O2 --> 2CO2 +3H2O + Heat
This reaction is called an exothermic reaction due to the evolution of heat.
c) when methane reacts with chlorine in the presence of light, the following reaction takes place:
It is considered a substitution reaction because in this reaction hydrogen is substituted with chlorine.
(a) Define the following terms in the context of spherical mirrors:
i) Pole
ii) Centre of curvature
iii) Principal axis
iv) Principal focus
b) Draw ray diagrams to show the principal focus of a
i) concave mirror
ii) convex mirror
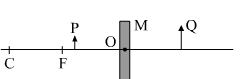
c) Consider the following diagram in which M is a mirror and P is an object and Q is its magnified image formed by the mirror.
(a) Write the functions of the following parts in human female reproductive system:
(i) Ovary
(ii) Oviduct
(iii) Uterus
(b) Describe the structure and function of placenta.
What is meant by speciation? List four factors that could lead to speciation. Which of them cannot be a major factor in the speciation of a self pollinating plant species. Give reason to justify your answer.
