 Multiple Choice Questions
Multiple Choice Questions18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100o C is
759.00 Torr
7.60 Torr
76.00 Torr
76.00 Torr
C.
76.00 Torr

HBr reacts with CH2 = CH – OCH3 under anhydrous conditions at room temperature to give
CH3CHO and CH3Br
BrCH2CHO and CH3OH
BrCH2 – CH2 – OCH3
H3C – CHBr – OCH3
The IUPAC name of the compound shown below is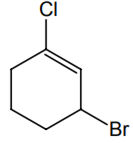
2-bromo-6-chlorocyclohex-1-ene
6-bromo-2-chlorocyclohexene
3-bromo-1-chlorocyclohexene
3-bromo-1-chlorocyclohexene
The increasing order of the rate of HCN addition to compounds A – D is
(A) HCHO
(B) CH3COCH3
(C) PhCOCH3
(D) PhCOPh
A < B < C < D
D < B < C < A
D < C < B < A
D < C < B < A
The term anomers of glucose refers to
isomers of glucose that differ in configurations at carbons one and four (C-1 and C-4)
a mixture of (D)-glucose and (L)-glucose
enantiomers of glucose
enantiomers of glucose
Phenyl magnesium bromide reacts with methanol to give
a mixture of anisole and Mg(OH)Br
a mixture of benzene and Mg(OMe)Br
a mixture of toluene and Mg(OH)Br
a mixture of toluene and Mg(OH)Br
The pyrimidine bases present in DNA are
cytosine and adenine
cytosine and guanine
cytosine and thymine
cytosine and thymine
Among the following the one that gives positive iodoform test upon reaction with I2 and NaOH is
CH3CH2CH(OH)CH2CH3
C6H5CH2CH2OH
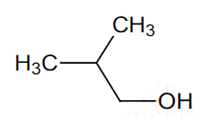

Fluorobenzene (C6H5F) can be synthesized in the laboratory
by heating phenol with HF and KF
from aniline by diazotisation followed by heating the diazonium salt with HBF4
by direct fluorination of benzene with F2 gas
by direct fluorination of benzene with F2 gas
The correct order of increasing acid strength of the compounds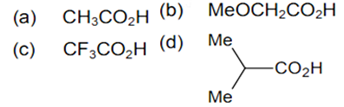
b < d < a < c
d < a < c < b
d < a < b < c
d < a < b < c
