 Multiple Choice Questions
Multiple Choice QuestionsBoron cannot form which one of the following anions?
BF63-
BH4-
B(OH)4-
B(OH)4-
A.
BF63-
because of the non-availability of d-orbitals, boron is unable to expand its octet. Therefore, the maximum covalence of boron cannot exceed 4.
The degree of dissociation (α) of a weak electrolyte ,AxBy is related to van't Hoff factor (i) by the expression
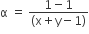
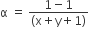
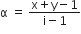
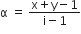
The presence or absence of hydroxy group on which carbon atom of sugar differentiates RNA and DNA.
1st
2nd
3rd
3rd
Phenol is heated with a solution of a mixture of KBr and KBrO3.The major product obtained in the above reaction is
2-Bromophenol
3-Bromophenol
4-Bromophenol
4-Bromophenol
Which of the following reagents may be used to distinguish between phenol and benzoic acid ?
Aqueous NaOH
Tollen's reagent
Molisch reagent
Molisch reagent
Trichloroacetaldehyde was subjected to Cannizzaro's reaction by using NaOH. The mixture of the products contains sodium trichloroacetate ion and another compound. The other compound is
2, 2, 2-Trichloroethanol
Trichloromethanol
2, 2, 2-Trichloropropanol
2, 2, 2-Trichloropropanol
The strongest acid amongst the following compounds is
CH3COOH
HCOOH
CH3CH2CH(Cl)CO2H
CH3CH2CH(Cl)CO2H
Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of,
two ethylenic double bonds
a vinyl group
an isopropyl group
an isopropyl group
Sodium ethoxide has reacted with ethanoyl chloride. The compound that is produced in the above reaction is
Diethyl ether
2-Butanone
Ethyl chloride
Ethyl chloride
Silver Mirror test is given by which one of the following compounds?
Acetaldehyde
Acetone
Formaldehyde
Formaldehyde
