 Multiple Choice Questions
Multiple Choice QuestionsWhat will be the decreasing order of stability of following carbocations?
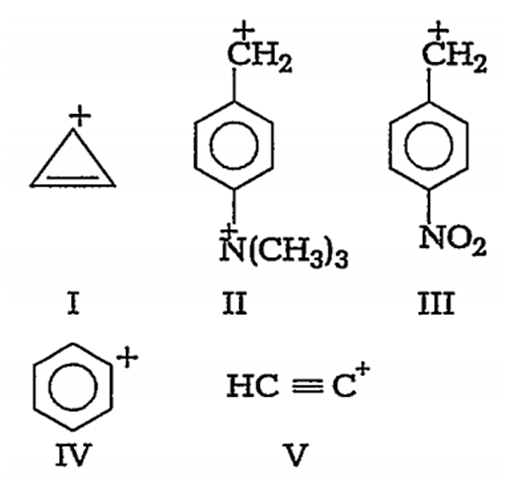
V >IV >III >II >I
I > II> III >IV >V
II >V > IV> I> II
I> II> III >V> IV
Milk is an emulsion in which
a gas is dispersed in water
lactose is dispersed in water
milk fat is dispersed in water
a solid is dispersed in water
For a non-volatile solute,
vapour pressure of solution is more than vapour pressure of solvent
Vapour pressure of solvent is zero
Vapour pressure of solvent is zero
All of the above
Which one is not the property of crystalline solid?
A definite and regular geometry
High intermolecular forces
Isotropic
Sharp melting point
Transition state 2 (TS2) is structurally most likely as a/an

intermediate 2
product
intermediate 1
transition state 3(TS3)
The decreasing order of electron affinity is
I > Br > Cl > F
Br > Cl > F > I
F > Cl > Br > I
Cl > F > Br > I
