 Multiple Choice Questions
Multiple Choice QuestionsThe osmotic pressure of which solution is maximum? (consider that decimormal solution of each is 90% dissociated) :
Aluminium sulphate
Barium chloride
Sodium sulphate
A mixture of equal volumes of (b) and (c)
A.
Aluminium sulphate
As the concentration and percentage dissociation is equal for each salt, hence the osmotic pressure will depend only on the number of ions produced. Hence osmotic pressure of Al2(SO4)3 will be the highest as it produce maximum ions (5).
Al2(SO4)3 2Al3+ + 3SO42-
What will be the product ,when carboxy phenol ,obtained by Reimer Tiemann's process ,is deoxidised with Zn powder ?
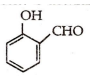
![]()
![]()
What is the correct IUPAC name of :
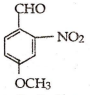
4 - methoxy - 2 - nitrobenzaldehyde
4 - formyl - 3 - nitro anisole
4 - methoxy - 6 - nitrobenzaldehyde
2 - formyl - 5 - methoxy nitrobenzene
The safest and the most common alternative of sugar is :
glucose
aspartame
saccharin
cyclodextrin
The reaction ,
CH3-CO-OCH3 + C2H5OH CH3-CO-OC2H5 + CH3OH is called :
Perkin's reaction
Claisen Schmidt reaction
Esterification
Trans-esterification
The product obtained ,heating ethanol with conc. H2SO4 at 165° - 170°C is :
(C2H5)2SO4
CH2=CH2
CH2F-CH2F
CF2=CFCl
