 Multiple Choice Questions
Multiple Choice QuestionsThe pair whose both species are used in antiacid medicinal preparation is
NaHCO3 and Mg(OH)2
Na2CO3 and Ca(HCO3)2
Ca(HCO3)2 and Mg(OH)2
Ca(OH)2 and NaHCO3.
A.
NaHCO3 and Mg(OH)2
Intestine is acidic due to formation of HCl during digestion. Excessive acidity in the intestine creates problem of intestinal ulcer, gastric reflux and oesophagitis. The compound which reduced or neutralise acidity are called antacids.
NaHCO3+ HCl NaCl+ H2O + CO2
Mg(OH)2 + 2HCl MgCl2 + 2H2O
The energy gaps (Eg) between valence band and conduction band for diamond, silicon and germanium are in the order
Eg (diamond) > Eg (silicon) > Eg (germanium)
Eg (diamond) <Eg (silicon) <Eg (germanium)
Eg (diamond) = Eg (silicon)= Eg (germanium)
Eg (diamond)> Eg (germanium)> Eg (silicon).
Among the following which one can have a meso form?
CH3CH(OH)CH(Cl)C2H5
CH3CH(OH)CH(OH)CH3
C2H5CH(OH)CH(OH)CH3
HOCH2CH(Cl)CH3
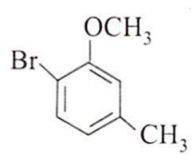
The major product obtained on the monobromination (with Br2/FeBr3) of the following compound A is
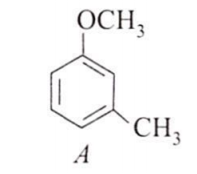
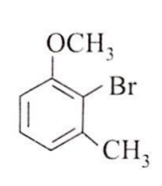
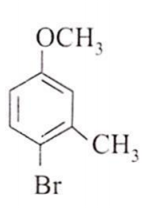
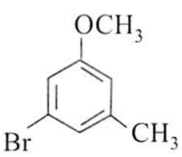
Assertion : [Co(NO2)3(NH3)3] does not show optical isomerism.
Reason : It has a plane of symmetry.
If both assertion and reason are true and reason is the correct explanation of the assertion.
If both assertion and reason are true but reason is not the correct explanation of the assertion.
If assertion is true, but reason is false.
Both assertion and reason are false statements.
Assertion: Silicones are hydrophobic in nature.
Reason: Si-O- Si linkages are moisture sensitive.
If both assertion and reason are true and reason is the correct explanation of the assertion.
If both assertion and reason are true but reason is not the correct explanation of the assertion.
If assertion is true, but reason is false.
Both assertion and reason are false statements.
Assertion: Diastereomers have different physical properties.
Reason : They are non-superimposable mirror images.
If both assertion and reason are true and reason is the correct explanation of the assertion.
If both assertion and reason are true but reason is not the correct explanation of the assertion.
If assertion is true, but reason is false.
Both assertion and reason are false statements.
Assertion: The presence ofnitro group facilitates nucleophilic substitution reactions in aryl halides.
Reason: The intermediate carbanion is stabilised due to the presence of nitro group.
If both assertion and reason are true and reason is the correct explanation of the assertion.
If both assertion and reason are true but reason is not the correct explanation of the assertion.
If assertion is true, but reason is false.
Both assertion and reason are false statements.
