 Multiple Choice Questions
Multiple Choice QuestionsLithium metal crystallises in a body centred cubic crystal. If the length of the side of the unit cell lithium is 351 pm, the atomic radius of the lithium will be
240.8 pm
151.8 pm
75.5
75.5
B.
151.8 pm
In case of body centred cubic (bcc) crystal,
Hence, atomic radius of lithium,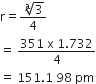
For the reaction, N2 + 3H2 → 2NH3, If 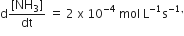 the value of
the value of 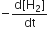 would be
would be
3 x 10-4 mol L-1 s-1
4 x 10-4 mol L-1s-1
6 x 10-4 mol L-1s-1
6 x 10-4 mol L-1s-1
Out of TiF62-, CoF63-, Cu2Cl2 and NiCl42- (Z of Ti = 22, Co= 27, Cu = 29, Ni =28) the colourless species are
TiF62- and CoF63-
Cu2Cl2 and NiCl42-
TiF62- and Cu2Cl2
TiF62- and Cu2Cl2
Which of the following compound will exhibit cis-trans (geometrical) isomerism?
2-butene
Butanol
2-butyne
2-butyne
The values of ΔH and ΔSfor the reaction, C(graphite) + CO2 → 2 CO (g) are 170 kJ and 170 JK-1 respectively. This reaction will be spontaneous at
710 K
910 K
1110 K
1110 K
In the reaction,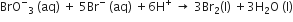
The rate of appearance of bromine (Br)2 is related to rate of disappearnace of bromide ions as following
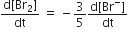

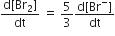
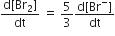
The stability of +1 oxidation state increases in the sequence
Al < Ga <In < Tl
Tl < In <Ga< Al
In < Tl < Ga < Al
In < Tl < Ga < Al
A 0.0020 m aqueous solution of an ionic compound Co(NH3)5(NO2)Cl freezes at -0.00732o C . Number of moles of ions which 1 mol of ionic compound produces on being dissolved in water will be (kf = - 1.86o C/m)
2
3
4
4
Which of the following reactions is an example of nucleophilic substitution reaction?
RX + KOH → ROH + KX
2RX + 2Na → R- R
RX + H2 → RH + HX
RX + H2 → RH + HX
Which one of the following is employed as a tranquilizer?
Equanil
Naproxen
Tetracycline
Tetracycline
