 Multiple Choice Questions
Multiple Choice QuestionsCobalt (III) chloride forms several octahedral complexes with ammonia, which of the following will not give a test for chloride ions with silver nitrated at 250 C?
CoCl. 3NH3
CoCl. 4NH3
CoCl. 5NH3
CoCl. 5NH3
A.
CoCl. 3NH3
a) [Co(NH3)3Cl3] --> [Co(NH3)2Cl3]
b) [Co(NH3)4Cl2]Cl --> [Co(NH3)4Cl2]+ +Cl-
c)[Co(NH3)5Cl]Cl2 --> [Co(NH3)5Cl]+ +2Cl-
d) [Co(NH3)6Cl]Cl2 --> [Co(NH3)6]+ +3Cl-
In the compound (a) does not ionise so does not give test for chloride ions.
The activation energy of a reaction can be determined from the slope of which of the following graphs?
InK vs T
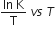


A mixture of gases contains H2 and O2 gases in the ratio of 1:4 (w/w).what is the molar ratio of the two gases in the mixture?
1:4
4:1
16:1
16:1
A given metal crystallises out with a cubic structure having edge length of 361 pm. If there are four metal atoms in one unit cell, what is the radius of one atom.
40 pm
127 pm
80 pm
80 pm
When initial concentration of a reactant is doubled in a reaction, its half-life period os not affected the order of the reaction is?
Zero
first
second
second
A device that converts energy of combustion of fuels like hydrogen and methane, directly into electrical energy is known as
fuel cell
electrolytic cell
dynamo
dynamo
The boiling point of 0.2 mol kg-1 solution of X in water is greater than the equimolal solution of Y in water. Which one of the following statements is true is this case?
X is undergoing dissociation in water
The molecular mass of X is greater than the molecular mass of Y.
Molecular mass of X is less than the molecular mass of Y,
Molecular mass of X is less than the molecular mass of Y,
Which one of the following electrolytes has the same value of van't Hoff's factor (i) as that of Al2(SO4)3 (if all are 100% ionised)?
K2SO4
K3[Fe(CN)6]
Al(NO3)3
Al(NO3)3
The correct bond order in the following species is
O22+ > O+2> O2-
O22+ < O+2< O2-
O22+ > O+2< O2-
O22+ > O+2< O2-
