 Multiple Choice Questions
Multiple Choice QuestionsHalf-lives of two radioactive elements A and B are 20 minutes and 40 minutes, respectively. Initially, the samples have equal number of nuclei. After 80 minutes, the ratio of decayed numbers of A and B nuclei will be:
1: 16
4 : 1
1: 4
1: 4
Proton, Deuteron and alpha particle of the same kinetic energy is moving in circular trajectories in a constant magnetic field. The radii of the proton, deuteron and alpha particle are respectively rp, rd and rα. Which one of the following relations is correct?
rα = rp= rd
rα = rp< rd
rα > rd> rp
rα > rd> rp
Assume that a neutron breaks into a proton and an electron. The energy released during this process is(Mass of neutron = 1.6725 x 10–27kg; mass of proton = 1.6725 x 10–27kg; mass of electron = 9 x 10–31kg)
0.73 MeV
7.10 MeV
6.30 MeV
6.30 MeV
The half life of a radioactive substance is 20 minutes. The approximate time interval (t2 - t1) between the time t2 when 2/3 of it has decayed and time t1 when 1/3 of it had decayed is
14 min
20 min
28 min
28 min
A nucleus of mass M + Δm is at rest and decays into two daughter nuclei of equal mass M/2 each. The speed of light is c.
The binding energy per nucleon for the parent nucleus is E1 and that for the daughter nuclei is E2. Then
E1 = 2E2
E2 = 2E1
E1 > E2
E1 > E2
A nucleus of mass M + Δm is at rest and decays into two daughter nuclei of equal mass M/2 each. Speed of light is c.
The speed of daughter nuclei is
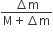



A radioactive nucleus (initial mass number A and atomic number Z) emits 3 α–particles and 2 positions. The ratio of number of neutrons to that of protons in the final nucleus will be
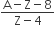
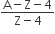
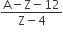
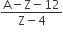
A radioactive nucleus A with a half-life T, decays into a nucleus B. At t = 0, there is no nucleus B. At some time t, the ratio of the number of B to that of A is 0.3. Then, t is given by
t = T log (1.3)
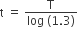
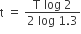
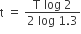
The above is a plot of binding energy per nucleon Eb, against the nuclear mass M; A, B, C, D, E, F correspond to different nuclei. Consider four reactions: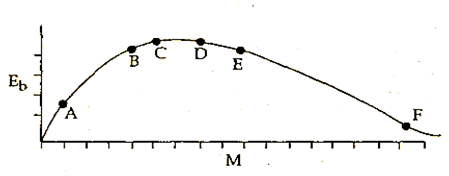
(i) A + B → C + ε (ii) C → A + B + ε
(iii) D + E → F + ε and (iv) F → D + E +ε
where ε is the energy released? In which reaction is ε positive?
(i) and (iv)
(i) and (iii)
(ii) and (iv)
(ii) and (iv)
The transition from the state n = 4 to n = 3 in a hydrogen-like atom results in ultraviolet radiation. Infrared radiation will be obtained in the transition from
2 → 1
3 → 2
4 → 2
4 → 2
