 Multiple Choice Questions
Multiple Choice QuestionsFor same mass of two different ideal gases of molecular weights M1 and M2, Plots of log V vs log p at a given constant temperature are shown. Identify the correct option.
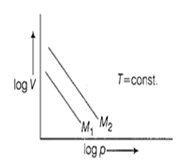
M1 > M2
M1 = M2
M1 < M2
Can be predicted only if temeperature is known
The pressure exerted by 6.0g of methane gas in a 0.03 m3 vessel at 129°C is (Atomic masses: C = 12.01, H = 1.01 and R= 8.314JK-1 mol-1)
215216 Pa
13409 Pa
41648 Pa
31684 Pa
C.
41648 Pa
Given,
volume, V = 0.03 m3
temperature,T = 129 + 273= 402 K
mass of methane, W = 6. 0 g
mol mass of methane, M = 12.01 + 4 x 1.01
= 16.05
From, ideal gas equation,
pV = nRT
p=
In clatherates of xenon with water, the nature of bonding between xenon and water molecule is
dipole induced dipole interaction
coordinate
hydrogen bonding
covalent
The vapour pressure of two liquids X and Y are 80 and 60 Torr respectively. The total vapour pressure of the ideal solution obtained by mixing 3 moles of X and 2 moles of Y would be
68 Torr
140 Torr
48 Torr
72 Torr
Which one of the following volume (V)- temperature (T) plots represents the behaviour of one mole of an ideal gas at one atmospheric pressure?
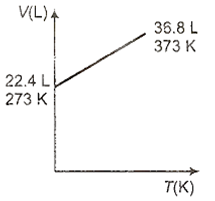
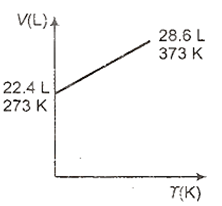
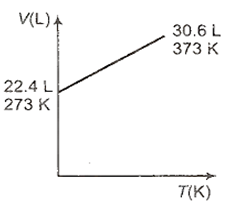
4 g of an ideal gas occupies 5.6035 L of volume at 546 K and 2 atmosphere pressure. What is its molecular weight?
4
16
32
64
If a gas contains only three molecules that move with velocities of 100, 200, 500 ms-1. What is the rms velocity of that gas in ms-1?
100
100√30
100√10
800/3
CH4 diffuses two times faster than a gas X. The number of molecules present in 32 g ofgas X is (N is Avogadro number)
N
N/2
N/4
N/16
Assertion (A): van der Waals' forces are reponsible for chemisorptions.
Reason (R): High temperature is favourable for chemisorptions.
The correct answer is:
(A) is false, but (R) is true
Both (A) and (R) are correct and (R) is the correct explanation of A
Both (A) and (R) are correct and (R) is not the correct explanation of (A)
(A) is true, but (R) is false
