 Multiple Choice Questions
Multiple Choice QuestionsFor same mass of two different ideal gases of molecular weights M1 and M2, Plots of log V vs log p at a given constant temperature are shown. Identify the correct option.
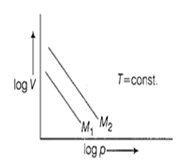
M1 > M2
M1 = M2
M1 < M2
Can be predicted only if temeperature is known
The pressure exerted by 6.0g of methane gas in a 0.03 m3 vessel at 129°C is (Atomic masses: C = 12.01, H = 1.01 and R= 8.314JK-1 mol-1)
215216 Pa
13409 Pa
41648 Pa
31684 Pa
In clatherates of xenon with water, the nature of bonding between xenon and water molecule is
dipole induced dipole interaction
coordinate
hydrogen bonding
covalent
The vapour pressure of two liquids X and Y are 80 and 60 Torr respectively. The total vapour pressure of the ideal solution obtained by mixing 3 moles of X and 2 moles of Y would be
68 Torr
140 Torr
48 Torr
72 Torr
D.
72 Torr
Moles of X, nX =
Moles of Y, nY =
pT = pXnX + pYnY
= 80 × + 60 ×
= 48 + 24 = 72 Torr
Therefore, 72 Torr is the total vapour pressure.
Which one of the following volume (V)- temperature (T) plots represents the behaviour of one mole of an ideal gas at one atmospheric pressure?
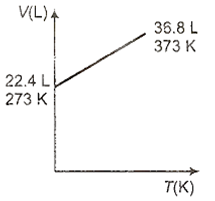
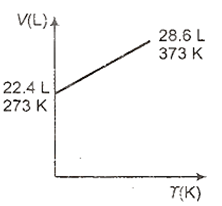
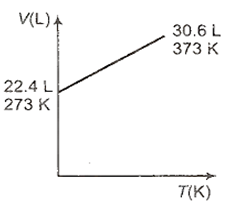
4 g of an ideal gas occupies 5.6035 L of volume at 546 K and 2 atmosphere pressure. What is its molecular weight?
4
16
32
64
If a gas contains only three molecules that move with velocities of 100, 200, 500 ms-1. What is the rms velocity of that gas in ms-1?
100
100√30
100√10
800/3
CH4 diffuses two times faster than a gas X. The number of molecules present in 32 g ofgas X is (N is Avogadro number)
N
N/2
N/4
N/16
Assertion (A): van der Waals' forces are reponsible for chemisorptions.
Reason (R): High temperature is favourable for chemisorptions.
The correct answer is:
(A) is false, but (R) is true
Both (A) and (R) are correct and (R) is the correct explanation of A
Both (A) and (R) are correct and (R) is not the correct explanation of (A)
(A) is true, but (R) is false
