 Multiple Choice Questions
Multiple Choice QuestionsWhich one will not show Cannizzaro reaction ?
Trimethyl acetaldehyde
Formaldehyde
Acetaldehyde
Benzaldehyde
The correct statement regarding a carbonyl compound with a hydrogen atom on its alpha -carbon, is
a carbonyl compound with a hydrogen atom on its alpha -carbon rapidly equilibrates with its corresponding enol and this process is known as aldehyde-ketone equilibration.
a carbonyl compound with a hydrogen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as carbonylation
a carbonyl compound with a hydrogen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as keto-enol tautomerism
a carbonyl compound with a hydrogen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as keto-enol tautomerism
Which of the following reagents would distinguish cis-cyclopenta-1-,2-diol from the trans isomer?
Ozone
MnO2
Aluminium isopropoxide
Aluminium isopropoxide
Treatment of cyclopentanone 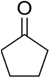
methyl lithium gives which of the following species?
Cyclopentanonyl anion
Cyclopentanonyl cation
Cyclopentanonyl radical
Cyclopentanonyl biradical
Given,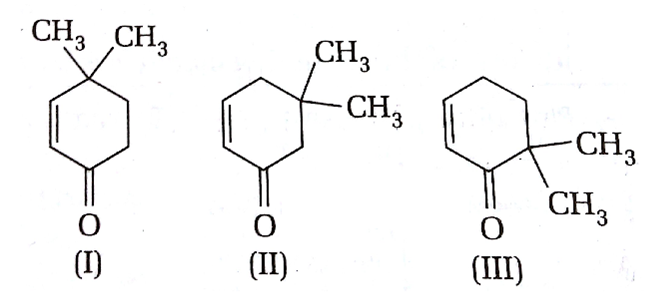
Which of the given compounds can exhibit tautomerism?
I and II
I and III
II and III
II and III
An organic compound X having molecular formula C5H10O yields phenyl hydrazone and gives negative response to the iodoform test and tollen test. It produces n -pentane on reduction X could be
pentanal
2-pentanone
3-pentanone
3-pentanone
The reaction by which benzaldehyde cannot be prepared?
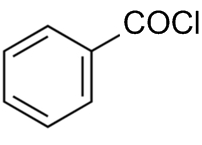 +H2 in presence of anhydrous AlCl3.
+H2 in presence of anhydrous AlCl3.
CH3CHO and C6H5CH2CHO can be distinguished chemically by
Benedict test
Iodoform test
Tollen's reagent test
Tollen's reagent test
B.
Iodoform test
CH3CHO and C6H5CH2CHO both being aliphatic aldehydes react with Tollen's reagent, Fehling solution and Benedict solution. So, these reagents cannot be used to distinguish them. CH3CHOreacts with NaOH and I2 to give yellow crystals of iodoform while C6H5CH2CHO does not react with it.
CH3CHO +3I2 4 NaOH → CHI3 +HCOONa +3NaI +3H2O
C6H5CH2CHO +I2 +4NaOH → No reaction
Thus, CH3CHO and C6H5CH2CHO can be distinguished by iodoform test.
