 Short Answer Type
Short Answer TypeAssign reasons for the following:
(i) Copper (I) ion is not known in aqueous solution.
(ii) Actinoids exhibit greater range of oxidation states than Lanthanoids.
Silver crystallizes in face-center cubic unit cell. Each side of this unit cell has a length of 400 pm. Calculate the radius of the silver atom. (Assume the atoms just touch each other on the diagonal across the face of the unit cell. That is each face atom is touching the four corner atoms.)
Nitrogen pentoxide decomposes according to equation: 2N2O5(g)---> 4NO2(g) + O2(g)
This first order reaction was allowed to proceed at 40° C and the data below were collected:
|
[N2O5] (M) |
Time (min) |
|
0.400 |
0.00 |
|
0.289 |
20.0 |
|
0.209 |
40.0 |
|
0.151 |
60.0 |
|
0.109 |
80.0 |
(a) Calculate the rate constant. Include units with your answer.
(b) What will be the concentration of N2O5 after 100 minutes?
(c) Calculate the initial rate of reaction.
Explain how the phenomenon of adsorption finds application in each of the following processes:
(i) Production of vacuum
(ii) Heterogeneous catalysis
(iii)Froth Floatation process
Describe the principle behind each of the following processes:
(i)Vapour phase refining of a metal.
(ii)Electrolytic refining of a metal.
(iii)Recovery of silver after silver ore was leached with NaCN.
(i) Vapour phase refining
Vapour phase refining is the process of refining metal by converting it into its volatile compound and then, decomposing it to obtain a pure metal. The basic principles involved in this process are:
(a) The metal should form a volatile compound with an available reagent, and
(b) The volatile compound should be easily decomposable so that the metal can be easily recovered.
Nickel, zirconium, and titanium are refined using this method.
(ii) Electrolytic refining is the process of refining impure metals by using electricity. In this process, impure metal is made the anode and a strip of pure metal is made the cathode. A solution of a soluble salt of the same metal is taken as the electrolyte. When an electric current is passed, metal ions from the electrolyte are deposited at the cathode as a pure metal and the impure metal from the anode dissolves into the electrolyte in the form of ions. The impurities present in the impure metal gets collected below the anode. This is known as anode mud.![]()
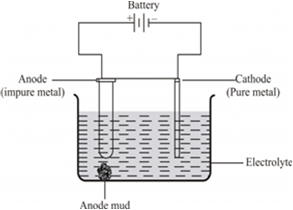
(iii) In the process of leaching, the finely divided silver is treated with a dilute solution of sodium cyanide while a current of air is continuously passed. As a result, silver passes into the solution.
forming sodium dicyanoargenate (I) while the impurities remain unaffected which are filtered off.
Ag2S + 4NaCN ---> 2Na [Ag(CN2)] + Na2S
Sodium dicyanoargenate (I)
Write the name, stereochemistry and magnetic behavior of the following:
(At. nos. Mn = 25, Co = 27, Ni = 28)
(i) K4[Mn (CN)6]
(ii) [Co (NH3)5Cl]Cl2
(iii) K2[Ni (CN)4]
