 Short Answer Type
Short Answer TypeUsing IUPAC norms write the formulate for the following coordination compounds:
(i) Hexaamminecobalt (III) chloride
(ii) Potassium tetrachloridonickelate (II)
Arrange the following in increasing order of their basic strength:
(i) C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
(i) C6H5-NH2 < C6H5-NH-CH3 < C6H5-CH2-NH2
Reason:
C6H5-NH2 will be least basic because of the delocalization of the lone pair of electrons present on the N-atom over the benzene ring due to the ‒R effect of the C6H5 group. However, C6H5-CH2-NH2 will be more basic than C6H5-NH-CH3 because of the electron-releasing nature of the CH3- a group that increases the electron density on the N-atom, making the lone pair of electrons on the N-atom easily available for donation to a proton. The basicity of C6H5-NH-CH3 will be intermediate of C6H5-NH2 and C6H5-CH2-NH2 because the C6H5- the group will tend to pull the electron density from the N-atom. On the other hand, the CH3- group will tend to increase the electron density on the N-atom. Thus, the basic strength of the given amines will follow the above-mentioned order.
(ii) 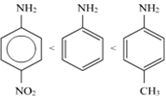
Reason:
The -CH3 group will increase the electron density on the benzene ring because of it is +I effect, while the NO2 group will decrease the electron density on the benzene ring because of its ‒I effect. Hence, the C6H5-NH2 molecule having - the CH3 group will be more basic than C6H5-NH2. Also, it will be more basic than the C6H5-NH2 molecule with the NO2 group.
Why does a solution containing non-volatile solute have higher boiling point than the pure solvent? Why is elevation of boiling point a colligative property?
(i) What is the principle behind the zone refining of metals?
(ii) What is the role of silica in the extraction of copper?
(iii) How is 'cast iron' different from 'pig iron'?
