 Short Answer Type
Short Answer TypeAccount for the yellow colour that appears in concentrated Nitric acid when it left standing in an ordinary glass bottle.
Which feature of the Ammonia molecule leads to the formation of the Ammonium ion when Ammonia dissolves in water?
Give one test that can be used to detect the presence of the ion produced in (b) (ii) .
Write the equations for the following reactions which result in the formation of Ammonia:-
1) A mixture of Ammonium chloride and slaked lime is heated.
2) Aluminium nitride and water.
The equations given below relate to the manufacture of sodium carbonate (molecular weight of Na2CO3 =106).
1. NaCl +NH3 +CO2 +H2O ---> NaHCO3 +NH4Cl
2. 2NaHCO3 ---> Na2CO3 +H2O +CO2
Question a and b are based on the production of 21.2 g of sodium carbonate.
a. what mass of sodium hydrogen carbonate must be heated to give 21.2 g of sodium carbonate (molecular weight of NaHCO3 =84)?
b. To produce the mass of sodium hydrogen carbonate calculated in (a), what volume of Carbon dioxide, measured at S.T.P., would be required?
a) 2 NaHCO3 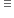 Na2CO3
Na2CO3
2 x 84 =168 106
For obtaining 106g of Na2CO3 amount of NaHCO3 heated = 168g.
therefore for obtaining 21.2 g of Na2CO3 amount of NaHCO3 heated = 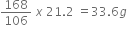
b) NaHCO3 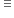 CO2
CO2
84g 22.4 litre
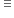 22.4 litre of CO2
22.4 litre of CO2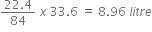
[Calcium, copper Lead, Aluminium, Zinc, Chromium, Magnesium, Iron.]
Choose the major metals from the list given above to make the following alloys:-
1) Stainless steel.
2) Brass
