 Multiple Choice Questions
Multiple Choice QuestionsThe equilibrium constants KP1 and KP2 for the reactions X ⇌2Y and Z ⇌ P + Q, respectively are in the ratio of 1 : 9. If the degree of dissociation of X and Z be equal then the ratio of total pressure at these equilibria is
1:1
1:36
1:9
1:9
Oxidising power of chlorine in aqueous solution can be determined by the parameters indicated below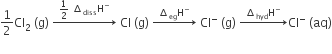
The energy involved in conversion of 
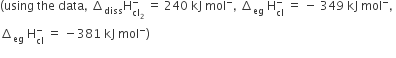
152 kJ mol-
-610 kJ mol-
-850 kJ mol-
-850 kJ mol-
For the following three reactions a, b and c, equilibrium constants are given
a. CO (g) + H2O (g) ⇌ CO2 (g)+ H2 (g); K1
b. CH4(g) + H2O (g) ⇌ CO (g) + 3H2 (g); K2
c. CH4(g) + 2H2O (g) ⇌ CO2 (g) + 4H2 (g); K2
Which of the following relations is correct?
K1√K2 = K3
K2K3 = K1
K3 = K1K2
K3 = K1K2
Standard entropy of X2, Y2 and XY3 are 60, 40 and 50 JK−1
mol −1, respectively. For the reaction,1/2X2 + 3/2Y2, ΔH = -30 kJ,to be at equilibrium, the temperature will be
1250 K
500 K
750 K
750 K
Four species are listed below
i. HCO3−
ii. H3O+
iii. HSO4−
iv. HSO3F
Which one of the following is the correct sequence of their acid strength?
iv < ii < iii < i
ii < iii < i < iv
i < iii < ii < iv
i < iii < ii < iv
Which one of the following pairs of species has the same bond order?
CN− and NO+
CN− and CN+
O2− and CN−
O2− and CN−
The ionization enthalpy of a hydrogen atom is 1.312 × 106Jmol−1. The energy required to excite the electron in the atom from n = 1 to n = 2 is
8.51 × 105 Jmol−1
6.56 × 105 Jmol−1
7.56 × 105 Jmol−1
7.56 × 105 Jmol−1
The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the corresponding salt, BA, will be
9.58
4.79
7.01
7.01
Identify the wrong statements in the following:
Chlorofluorocarbons are responsible for ozone layer depletion
Greenhouse effect is responsible for global warming
Ozone layer does not permit infrared radiation from the sun to reach the earth
Ozone layer does not permit infrared radiation from the sun to reach the earth