 Multiple Choice Questions
Multiple Choice QuestionsWhich one of the following order is correct for the first ionisation energies of the elements?
B <Be <N < O
Be <B <N < O
B <Be <O <N
B <O <Be <N
The cubic unit cell of a metal (molar mass = 63.55 g mol-1) has an edge length of 362 pm. Its density is 8. 92 g cm-3. The type of unit cell is
primitive
face centred
body centred
end centred
The equilibrium constant for the given reaction is 100.
What is the equilibrium constant for the reaction given below?
10
1
0.1
0.01
20 mL of 0.1 M acetic acid is mixed with 50 mL of potassium acetate. Ka of acetic acid = 1.8 x 10-5 at 27C. Calculate concentration of potassium acetate if pH of the mixture is 4.8.
0.1 M
0.04 M
0.4 M
0.02 M
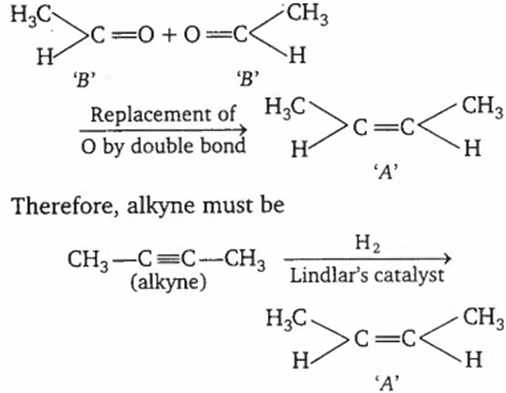
Identify the alkyne in the following sequence of reactions,
A.
In Wacker process, alkene is oxidised into aldehyde.
Since on ozonolysis, only alkenes produce aldehydes, 'A' must be an alkene. To decide the structure of alkene that undergoes ozonolysis, bring the products together in such a way that O atoms are face to face and, replace O by double (=) bond. Thus,
Match the following.
| List I | List II | ||
| (A) | Flespar | (I) | [Ag3SbS3] |
| (B) | Asbestos | (II) | Al2O3.H2O |
| (C) | Pyrargyrite | (III) | MgSO4.H2O |
| (D) | Diaspore | (IV) | KAlSi3O8 |
| (V) | CaMg3(SiO3)4 |
(A) (B) (C) (D)
IV V II I
(A) (B) (C) (D)
IV V I II
(A) (B) (C) (D)
IV I III II
(A) (B) (C) (D)
II V IV I
The concentration of an organic compound in chloroform is 6.15 g per 100 mL of solution. A portion of this solution in a 5 cm polarimeter tube causes an observed rotation of -1.2°. What is the specific rotation of the compound?
+12
-3.9
-39
+61.5
Fluorine reacts with dilute NaOH and forms a gaseous. product A. The bond angle in the molecule of A is
104°40'
103°
107°
109°28'
