 Multiple Choice Questions
Multiple Choice QuestionsTo an aqueous solution containing anions a few drops of acidified KMnO4 are added. Which one of the following anions, if present will not decolourise the KMnO4 solution ?
I-
CO
S2-
NO
In the following reaction,
Mx+ + MnO → MO + Mn2+ + O2
If one mole of MnO oxidises 2.5 moles of Mx+, then the value of x is
5
3
2
1
0.25 g of an organic compound on Kjeldahl's analysis gave enough ammonia to just neutralize 10 cm3 of 0.5 M H2SO4. The percentage of nitrogen in the compound is
28
56
14
112
Lassaigne's test for the detection of nitrogen fails in
H2N-CO-NHNH2.HCl
NH2-NH2.HCl
NH2-CO-NH2
C6H5-NH-NH2.HCl
When tetrahydrafuran is treated with excess HI, the product formed is
1, 4-diiodobutane
1,4-butanediol
2-iodotetrahydrofuran
4-iodo-1-butanol
Pick out the correct statements from the following and choose the correct answer from the codes given below
1. Hexa-1, 5-diene is a conjugated diene
2. Prop-1, 2-diene is conjugated diene
3. Hexa-1, 3-diene is a conjugated diene
4. Buta-1, 3-diene is an isolated diene
5. Prop-1, 2-diene is a cumulative diene
1, 2
3, 5
2, 3
4, 5
B.
3, 5
In conjugated diene alternate single and double bonds are present while in cumulative diene, double bonds are present at adjacent positions.
1. Hexa-1, 5-diene is an isolated diene.
2. Prop-1,2 -diene is a cumulative diene.
3. Hexa-1, 3-diene is a conjugated diene.
4. Buta-1, 3-diene is a conjugated diene.
5. Prop-1, 2-diene is a cumulative diene.
Hence, among all the statements, statement 3 and 5 are correct.
The correct IUPAC name of the acid
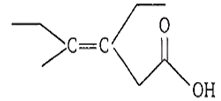
Z-3-ethyl-4methyl hex-3-en-1-oic acid
E-3-ethyl-4-ethylhex-3-en-1-oic acid
Z-3-ethyl-4-methyl hexanoic acid
Z-3, 4-diethylpent-3-en-1-oic acid
The elements present in the core of earth are collectively known as
lithophiles
nucleophiles
chalcophiles
siderophiles
