 Multiple Choice Questions
Multiple Choice QuestionsOctane number of petrol can be improved by admixing it with which of the following chemicals?
Et4Pb
CH3OC(CH3)3
Both (a) and (b)
Pyrene
Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3-trimethyl cyclopropane are examples of which one of the following?
Constitutional isomers
Structural isomers
Both (a) and (b)
Structural as well as positional isomers
How many structural isomers are possible for a molecule of C6H12,composition but having at least-one carbocyclic ring?
Seven
Six
Five
None of these
The configurations of the chiral centres in A and B are, respectively, as
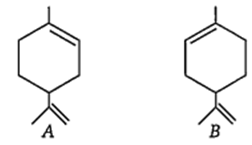
R and S
S and R
R and R
S and S
Arrange Na+, Mg+ and Al3+ in increasing order of energy of hydration.
Na+ < Mg+ < Al3+
Al3+< Mg+ < Na+
Al3+ < Na+ < Mg2+
Mg2+< Al3+ < Na+
Which one of K, I, Cl and Li will display the highest first ionisation energy?
K
I
Cl
Li
C.
Cl
The ionisation energy increases on moving from left to right along a period, because with increase in atomic number in a period, effective nuclear charge increases and so, electrons are more tightly held. Whereas, on moving down a group, the valence shell becomes far away from the nucleus and thus, nuclear attraction towards valence electron decreases which results in-decrease in ionisation energy. Thus, chlorine has the highest first ionisation energy among all the given elements.
You may have noticed that chicken-egg is very strong along its long axis so much so that it does not break even when pressed very hard. Which of the following is the main constituent of egg-shell?
CaSO4·2H2O
CaSO4·1/2H2O
CaSiO3
CaCO3
Nuclear attraction is often the deciding control factor for the association of neutral molecules to a given metal ion. Which one of the following represents the correct order of stability of the ions?
[Be(H2O)4]2+, [Mg(H2O)4]2+, [Ca(H2O)4]2+ and [Sr(H2O)4]2+ ?
[Be(H2O)4]2+ > [Sr(H2O)4]2+> [Mg(H2O)4]2+ > [Ca(H2O)4]2+
[Ca(H2O)4]2+> [Mg(H2O)4]2+ > Be(H2O)4]2+ > [Sr(H2O)4]2+
[Sr(H2O)4]2+ > [Ca(H2O)4]2+> [Mg(H2O)4]2+ > Be(H2O)4]2+
[Be(H2O)4]2+> [Mg(H2O)4]2+ > [Ca(H2O)4]2+ > [Sr(H2O)4]2+
Which of the following represents correct acidity order Li2O , BeO and B2O3 ?
Li2O < BeO < B2O3
B2O3 < BeO < Li2O
BeO < Li2O < B2O3
BeO < B2O3 < Li2O
Which of the following does not/do not represent the correct equivalent mass of Cr2O in the reaction Cr2O+ 14 H+ + 6e- → 2Cr3+ + 7H2O.
The molar mass of dichromate
One third of the molar mass of dichromate
One half of the molar mass of dichromate
all of the above
