 Multiple Choice Questions
Multiple Choice Questions
The product Y is
p-chloro nitrobenzene
o-chloro nitrobenzene
m-chloro nitrobenzene
o, p-dichloro nitrobenzene
Following compounds are respectively ..... geometrical isomers
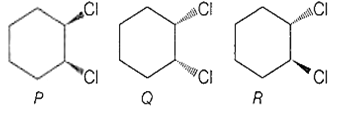
P - cis ;Q - cis ; R - trans
P - cis ; Q - trans ; R - trans
P - trans ; Q - cis ; R - cis
P - cis ; Q - trans ; R - cis
Which is more basic oxygen in an ester?
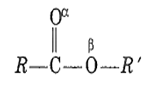
Carbonyl oxygen, α
Carboxyl oxygen, β
Equally basic
Both are acidic oxygen
Match the following and choose the correct option given below:-
| Compound / Type | Use |
| A. Dry ice | I. Anti-knocking compound |
| B. Semiconductor | II. Electronic diode or triode |
| C. Solder | III. Joining circuits |
| D. TEL | IV. Refrigerant for preserving food |
A - I; B - II; C - IV; D - III
A - II; B - III; C - I ; D - IV
A - IV; B - III; C - II; D - I
A - IV; B - II; C - III; D - I
B can be obtained from halide by van- Arkel method. This involves. This involves reaction
2BI3 2B + 3I2
2BCl3 + 3H2 2B +6HCl
Both (a) and (b)
None of the above
The density of solid argon is 1.65 g per cc at -233°C. If the argon atom is assumed to be a sphere of radius 1.54 × 10-8 cm, what percent of solid argon is apparently empty space? (Ar = 40)
16.5%
38%
50%
62%
Acid hydrolysis of ester is first order reaction and rate constant is given by
k =
where, V0, Vt and V∞ are the volume of standard NaOH required to neutralise acid present at a given time, if ester is 50% neutralised then
V∞ = Vt
V∞ = (Vt - V0)
V∞ = 2Vt - V0
V∞ = 2Vt + V0
Which of these ions is expected to be coloured in aqueous solution?
I. Fe3+
II. Ni2+
III. Al3+
I and II
II and III
I and III
I, II and III
A.
I and II
Among the given ions, Fe3+ and Ni2+ are expected to be coloured in aqueous solution as Fe3+ has 4 unpaired electrons whereas Ni2+ has 2 unpaired electrons. Al3+ has no unpaired elctrons in 3d, thus it is a colourless ion.
Consider the following reaction,

The reaction is of first order in each diagram, with an equilibrium constant of 104. For the conversion of chair form to boat form = 4.5 × 10-8 m at 298 K with pre-exponential factor of 1012 s-1. Apparent rate constant (=kA/ kB) at 298 K is
4.35 × 104 s-1
4.35 × 108 s-1
4.35 × 10-8 s-1
4.35 × 1012s-1
