 Multiple Choice Questions
Multiple Choice Questions1 mole of gas occupying 3 L volume is expanded against a constant external pressure of 1 atm to a volume of 15 L. The work done by a system is equal to
-1.215 × 103 J
-12.15 × 103 J
+1.215 × 103 J
+12.15 × 103 J
A.
-1.215 × 103 J
pext = 1 atm
V1 = 3L ⇒ V2 = 3L
As work is done against constant external pressure, the process is irreversible.
W = -pext V
= -1atm [15 - 3] L = -12atmm L
= -12 × 101.3 J (Since, 11 atm= 101.3 J)
= -1215.6 J
= -1.2156 × 103 J
The value for Cr, Mn, Fe and Co are -0.41, +0.77 and +1.97 V respectively. For which one of these metals the change in oxidation state from +2 to +3 is easiest?
Cr
Mn
Fe
Co
The oxidation numbers of phosphorus in Ba(H2PO2) and xenon in Na4XeO6 respectively are
+3 and +4
+2 and +6
+1 and +8
-1 and -6
Setting of plaster of Paris is
dehydration
oxidation with atmospheric oxygen
combination with atmospheric CO2
hydration to yield another hydrate
In the following compound, groups designated as A, B, C will be numbered as
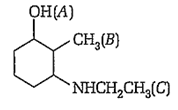
3, 2, 1
6, 1, 2
3, 1, 2
1, 2, 3
The following compound can exhibit
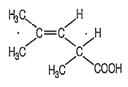
geometrical isomerism
geometrical and optical isomerism
optical isomerism
tautomerism
The IUPAC name for
CH3CH=CHCH2C(NH2)H-CH2-COOH
5-amino-2-heptenoic acid
5-amino hex-2-ene-carboxylic aod
3-amino-5-heptenoic acid
β-amino-δ-heptenoic acid
Petroleum is obtained from water gas, name of the reaction involved is
Fischer-Tropsch
Benzoic
Dow's
Kjeldahl's
Which of the following is correct increasing order for the value of ?
e < p < n < α
n < p < e < α
n < p < α < e
n < α < p < e
