 Multiple Choice Questions
Multiple Choice Questions1 mole of gas occupying 3 L volume is expanded against a constant external pressure of 1 atm to a volume of 15 L. The work done by a system is equal to
-1.215 × 103 J
-12.15 × 103 J
+1.215 × 103 J
+12.15 × 103 J
The value for Cr, Mn, Fe and Co are -0.41, +0.77 and +1.97 V respectively. For which one of these metals the change in oxidation state from +2 to +3 is easiest?
Cr
Mn
Fe
Co
The oxidation numbers of phosphorus in Ba(H2PO2) and xenon in Na4XeO6 respectively are
+3 and +4
+2 and +6
+1 and +8
-1 and -6
Setting of plaster of Paris is
dehydration
oxidation with atmospheric oxygen
combination with atmospheric CO2
hydration to yield another hydrate
In the following compound, groups designated as A, B, C will be numbered as
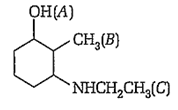
3, 2, 1
6, 1, 2
3, 1, 2
1, 2, 3
The following compound can exhibit
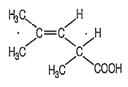
geometrical isomerism
geometrical and optical isomerism
optical isomerism
tautomerism
The IUPAC name for
CH3CH=CHCH2C(NH2)H-CH2-COOH
5-amino-2-heptenoic acid
5-amino hex-2-ene-carboxylic aod
3-amino-5-heptenoic acid
β-amino-δ-heptenoic acid
Which is not an electrophile?
H5
H3
BF3
SO3
A.
H5
H5has eight electrons (octet complete) on carbon. Thus, H5 is not an electrophile.
Petroleum is obtained from water gas, name of the reaction involved is
Fischer-Tropsch
Benzoic
Dow's
Kjeldahl's
Which of the following is correct increasing order for the value of ?
e < p < n < α
n < p < e < α
n < p < α < e
n < α < p < e
