 Multiple Choice Questions
Multiple Choice QuestionsAn element has a body centered cubic structure with a unit cell edge length of 400 pm. Atomic mass of an element is 24 g mol-1. What is the ·density of the element? (NA = 6 × 1023 mol-1)
2.50 g cm-3
1.80 g cm-3
3.60 g cm-3
1.25g cm-3
Which one of the following is more reactive towards SN2 reaction ?
(CH3)3CX
(CH3)2CHX
CH3CH2X
CH3X
Which one of the following is not used as an initiator in ionic polymerisation?
NaNH2
SnCl2
AlCl3
(C6H5CO)2O2
What are X and Y in the following reactions?
| X | Y |
| CH3-CH(CH3)-MgBr | C2H5COCH3 |
| X | Y |
| CH3CH2CH2MgBr | CH3-CO-CH3 |
| X | Y |
| (CH3)3CMgBr | CH3-CO-CH3 |
| X | Y |
| CH3-CH2-MgBr | CH3CH2CHO |
Which of the following structure represents the compound, generally added to soaps to impart antiseptic properties?
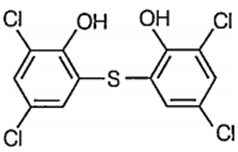
![]()
![]()
![]()
A.

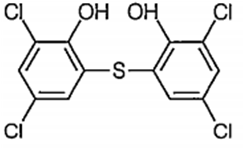
Is added and generally used to impart antiseptic properties in soap.
The increasing order of acidity of the following carboxylic acids is
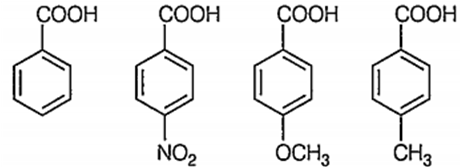
I II III IV
III < IV < II < I
II < III < II < I
I < II < IV < III
III < IV < I < II
Identify the statement which is not correct from the following.
(i)Carbohydrates are stored as glycogen in animals.
(ii) In glycylalanine, CO of peptide bond belongs to alanine.
(iii) Base - sugar- phosphate unit is known as nucleoside.
(iv) Obesity is due to hypothyroidism.
The correct answer is:
i, iv
ii, iii, iv
i, iii, iv
ii, iii
The drug, which was designed to prevent the interaction of histamine with the receptors present in the stomach wall is
prontosil
cimetidine
aspartame
equanil
