 Multiple Choice Questions
Multiple Choice QuestionsMnO (1 mole) in neutral aqueous medium is disproportionated to
2/3 mole of MnO and 1/3 mole of MnO2
1/3 mole of MnO and 2/3 mole of MnO2
1/3 mole of Mn2O7 and 1/3 mole of MnO2
2/3 mole of Mn2O7 and 1/3 mole of MnO2
Among the following the strongest acid is
CH3COOH
C6H5COOH
m-CH3OC6H4COOH
p-CH3OC6H4COOH
C.
m-CH3OC6H4COOH
Among all the given options, strongest acid is m-CH3OC6H4COOH.
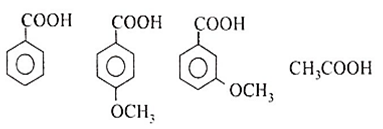
-OCH3 group displays electron withdrawing acid strengthening inductive effect at the meta position but at the para position, an electron releasing acid weakening resonance effect operates (which at this position outweighs the inductive effect).
Which of the following are arranged in the decreasing order of dipole moment?
CH3Cl, CH3Br, CH3F
CH3Br, CH3Cl, CH3F
CH3Cl, CH3Br, CH3F
CH3Br, CH3F, CH3Cl
Which of the following compounds possesses the C-H bond with the lowest bond dissociation energy?
Toluene
Benzene
n-Pentane
2,2-Dimethylpropane
One gram sample of NH4NO3 is decomposed in a bomb calorimeter. The temperature of the calorimeter increases by 6.12 K. The heat capacity of the system is 1.23 kJ/g/deg. What is the molar heat of decomposition for NH4NO3 ?
-7.53 kJ/mol
-398.1 kJ/mol
-16.1 kJ/mol
-602 kJ/mol
Which one of the following has S° greater than zero?
CaO(s) + CO2(g) CaCO3(s)
NaCl(aq) NaCl(s)
NaNO3(s) Na+(aq) + NO3-(aq)
N2(g) + 3H2(g) 2NH3(g)
