 Multiple Choice Questions
Multiple Choice QuestionsDimethyl glyoxime gives a red precipitate with Ni2+, which is used for its detection. To get this precipitate readily the best pH range is
< 1
2-3
3- 4
9- 11
D.
9- 11
Nickel(II) forms a precipitate with the organic compound dimethylglyoxime, C4H6(NOH)2. The formation of the red chelate occurs quantitatively in a solution in which the pH is buffered in the range of 5 to 9. The chelation reaction that occurs is illustrated below.
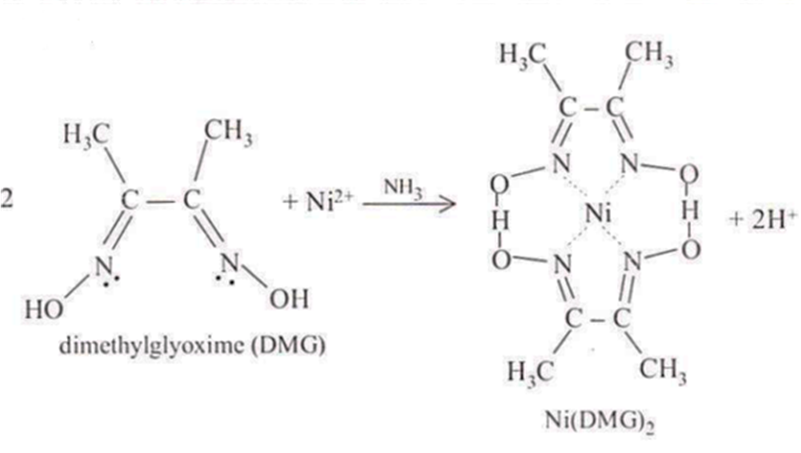
The reaction is performed in a solution buffered by either an ammonia or citrate buffer to prevent the pH of the solution from falling below 5. If the pH does become too low the equilibrium of the above reaction favors the formation of nickel(II) ion, causing the dissolution of Ni(DMG)2 back into the mother liquor.
The statement true for is
it has a non-linear structure
it is called pseudohalogen
the formal oxidation state of nitrogen in this anion is -1
it is isoelectronic with NO2
The compound insoluble in water is
mercurous nitrate
mercuric nitrate
mercurous chloride
mercurous perchlorate.
How much energy is released when 6 moles of octane is burnt in air? Given for CO2(g) , H2O(g) and C8H18(l) respectively are - 490, -240 and +160 kJ/mol.
-6.2 kJ
-37.4 kJ
-35.5 kJ
-20.0 kJ
For the equilibrium at 1 atm and 298K.
standard free energy change is equal to zero (= 0)
free energy change is less than zero ( < 0)
standard free energy change is less than zero (< 0)
standard free energy change is greater than zero (G° > 0).
