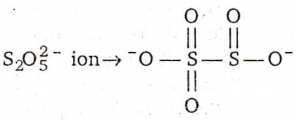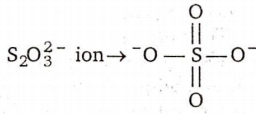Multiple Choice Questions
Multiple Choice QuestionsIf the Zn2+/Zn electrode is diluted to 100 times then the change in emf :
increase of 59 mV
decrease of 59 mV
increase of 29.5 mV
decrease of 29.5 mV
If equivalent conductance of 1 M benzoic acid is 12.8 ohm-1 cm and if the conductance of benzoate ion and H+ ion are 42 and 288 .42 ohm-1 cm2 respectively .Its degree of dissociation is :
39 %
3.9 %
0.35 %
0.039 %
If two substances A and B have : = 1 : 2 and have mole fraction in solution 1 : 2 then mole fraction of A in vapours :
0.33
0.25
0.52
0.2
If an aqueous solution of glucose is allowed to freeze then crystal of which will be separated out first ?
Glucose
Water
Both (a) and (b)
None of these
A + 2B C + D , if
- = 5 x 10-4 mol L-1 s-1 , then - is
2.5 x 10-4 mol L-1 s-1
50 x 10-4 mol L-1 s-1
2.5 x 10-3 mol L -1 s-1
1.0 x 10-3 mol L -1 s-1
Which statement is wrong for NO ?
It is anhydride of nitrous acid
Its dipole moment is 0.22 D
It forms dimer
It is paramagnetic
There is no S-S bond in :
S2O42-
S2O52-
S2O32-
S2O72-
D.
S2O72-
In S2O72- ion the two sulphur atoms are linked through an oxygen atom .Each sulphur atom is further-linked with oxygen atoms .Its structure is :
![]()
In all other ions S-S bond is present
![]()