 Multiple Choice Questions
Multiple Choice QuestionsIn the electronic structure of H2SO4 , the total number of unshared electrons is
20
16
12
8
A.
20
The structure of H2SO4 is
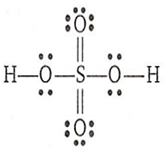
Total number of unshared electrons = 10 x 2 = 20
Therefore, among all the given options, option a is correct.
The correct order towards bond angle is
sp3 < sp2 < sp
sp < sp2 < sp3
sp < sp3 < sp2
sp2 < sp3 < sp
Which of the following relation is correct ?
Ist IE of C > Ist IE of B
Ist IE of C < Ist IE of B
IInd IE of C > IInd IE of B
Both (b) and (c)
Which of the following configurations corresponds to element of highest ionisation energy?
1s2, 2s1
1s2, 2s2, 2p3
1s2, 2s2, 2p2
1s2, 2s2, 2p6, 3s1
Which of the following has a bond formed by overlap of sp-sp3 hybrid orbitals?
CH3 - C C - H
CH3 - CH = CH - CH3
CH2 = CH - CH = CH2
HC CH
The density of a gas is found to be 1.56 g/L at 745 mm pressure and 65°C. What is the moleculer mass of the gas?
44.2 u
4.42 u
2.24 u
22.4 u
Considering H2O as weak field ligand, the number of unpaired electrons in [Mn(H2O)6] will be (Atomic number of Mn = 25)
five
two
four
three
The quantum numbers + and - for the electron spin represent
rotation of the electron in clockwise and anticlockwise direction respectively
rotation of the electron in anticlockwise and clockwise direction respectively
magnetic moment of the electron pointing up and down respectively
two quantum mechanical spin states which have no classical analogues
In long form of Periodic Table, the properties of the elements are a periodic function of their
atomic size
ionisation energy
atomic mass
atomic number
